Method Article
Isolation of Exosome-Enriched Extracellular Vesicles Carrying Granulocyte-Macrophage Colony-Stimulating Factor from Embryonic Stem Cells
W tym Artykule
Podsumowanie
This study describes a method to isolate exosome-enriched extracellular vesicles carrying immune-stimulatory granulocyte macrophage colony-stimulating factors from embryonic stem cells.
Streszczenie
Embryonic stem cells (ESCs) are pluripotent stem cells capable of self-renewal and differentiation into all types of embryonic cells. Like many other cell types, ESCs release small membrane vesicles, such as exosomes, to the extracellular environment. Exosomes serve as essential mediators of intercellular communication and play a basic role in many (patho)physiological processes. Granulocyte-macrophage colony-stimulating factor (GM-CSF) functions as a cytokine to modulate the immune response. The presence of GM-CSF in exosomes has the potential to boost their immune-regulatory function. Here, GM-CSF was stably overexpressed in the murine ESC cell line ES-D3. A protocol was developed to isolate high-quality exosome-enriched extracellular vesicles (EVs) from ES-D3 cells overexpressing GM-CSF. Isolated exosome-enriched EVs were characterized by a variety of experimental approaches. Importantly, significant amounts of GM-CSF were found to be present in exosome-enriched EVs. Overall, GM-CSF-bearing exosome-enriched EVs from ESCs might function as cell-free vesicles to exert their immune-regulatory activities.
Wprowadzenie
ESCs are derived from the blastocyst stage of a preimplantation embryo1. As pluripotent stem cells, ESCs have the capability to self-renew and differentiate into any type of embryonic cell. Due to their remarkable developmental potential and long-term proliferative capacity, ESCs are extremely valuable for biomedical research1. Current research efforts have largely focused on the therapeutic potential of ESCs for a variety of major pathological disorders, including diabetes, heart disease, and neurodegenerative diseases2,3,4.
Mammalian cells, including ESCs, are known to release vesicles with variable sizes to the extracellular environment, and these EVs possess many physiological and pathological functions due to their role in intercellular communication5. Among different subtypes of EVs, exosomes are small membrane vesicles released from various cell types into the extracellular space upon fusion of intermediate endocytic compartments, multivesicular bodies (MVBs), with the plasma membrane6. Exosomes have been reported to mediate intercellular communication and are critically involved in many (patho)physiological processes7,8. Exosomes inherit some biological functions from their own parental cells, because exosomes contain biological materials acquired from the cytosol, including proteins and nucleic acids. Thus, the associated antigens or factors stimulating the immune response specific for a given disease are encapsulated in the exosomes from particular types of cells9. This paved the way for clinical trials exploring tumor-derived exosomes as an anti-cancer vaccine10.
GM-CSF is a cytokine secreted by different types of immune cells11. Emerging evidence demonstrates that GM-CSF activates and regulates the immune system and plays an essential role in the antigen-presenting process12. For instance, a clinical report suggests that GM-CSF stimulates the immune response to tumors as a vaccine adjuvant13. Several GM-CSF-based cancer immunotherapy strategies to exploit the potent immune-stimulatory activity of GM-CSF have been investigated in clinical trials14. Among these, a cancer vaccine composed of irradiated GM-CSF-secreting tumor cells has shown some promise in advanced melanoma patients by inducing cellular and humoral antitumor responses and subsequent necrosis in metastasized tumors15.
Because the exosomes derived from ESCs possess similar biological activities as the original ESCs, maybe GM-CSF-carrying exosomes from ESCs could function as cell-free vesicles to regulate the immune response. In this paper, a detailed method to produce high-quality exosome-enriched EVs from ESCs expressing GM-CSF is described. These exosome-enriched EVs have the potential to serve as immune-regulatory vesicles to modulate the immune response.
Protokół
1. Culturing ES-D3 cells
- To generate exosome-free fetal bovine serum (FBS), load FBS into an ultracentrifuge and centrifuge at 100,000 x g for 16 h at 4 °C. Following centrifugation, collect serum supernatant as exosome-free FBS for culturing the murine ESC cell line ES-D3 and acquiring exosome-enriched EVs.
- Before plating the ES-D3 cells, coat 15 cm tissue culture dishes using gelatin (0.1%) at room temperature for 30 min.
- Following a previously described protocol16, culture the ES-D3 cells without feeder layer cells in the gelatin-coated 15 cm tissue culture dishes. The ES-D3 cell culture medium is composed of DMEM, exosome-free FBS (15%), nonessential amino acids (0.1 mM), L-glutamine (2 mM), β-mercaptoethanol (0.1 mM), penicillin (50 units/mL), streptomycin (50 µg/mL), and leukemia inhibitory factor (LIF; 100 units/mL). Culture ES-D3 cells at 37 °C in a 5% CO2 humidified incubator.
- Once the ES-D3 cells reach around 90% confluency in 15 cm tissue culture dishes, remove the medium by aspiration. Wash the cells using trypsin (5 mL; 0.05%). Add trypsin (5 mL) to the dishes and incubate at 37 °C for 5 min. Collect the cells from the dishes.
- Add fresh culture medium (5 mL) to the collected cells to inactivate the trypsin. Centrifuge the cells at 390 x g for 5 min. Resuspend the cells in fresh medium and determine the cell number using a hemocytometer.
- For passaging cells, plate the ES-D3 cells (5 x 106) in a gelatin-coated (0.1%) 15 cm tissue culture dish with fresh medium (15 mL) and culture for 3 days before subculturing the cells.
- To collect the cell culture supernatant for isolation of exosome-enriched EVs, plate the ES-D3 cells (1 x 107) in a gelatin-coated (0.1%) 15 cm tissue culture dish with fresh medium (15 mL) for 3 days prior to collecting cell culture supernatant.
2. Generation of GM-CSF expression plasmid
NOTE: Generate the transfection plasmid pEF1α-mGM-CSF-IRES-hrGFP to overexpress GM-CSF in ES-D3 cells. In this plasmid, expression of murine GM-CSF cDNA along with the marker protein humanized Renilla reniformis GFP (hrGFP) is driven by the human polypeptide chain elongation factor 1α (EF1α) promoter17,18.
- Generate the vector backbone.
- Digest the plasmid pEF1α-FD3ER-IRES-hrGFP (20 µg of DNA) using the restriction enzyme EcoRI (100 units) at 37 °C for 2 h to generate two DNA fragments: the vector backbone (6.0 kb) and FD3ER insert (2.5 kb).
- Transfer 50% of digested plasmid DNA (10 µg) into one 1.5 mL microcentrifuge tube and treat with alkaline phosphatase (20 units) at 37 °C for 1 h. Resolve both untreated and dephosphorylated DNA using agarose gel electrophoresis (2%).
- Purify the vector backbone DNA fragments (6.0 kb) using a DNA gel extraction kit . While the untreated vector backbone will serve as the empty vector (pEF1α-IRES-hrGFP), the dephosphorylated vector backbone will be used to generate GM-CSF-expressing plasmid (pEF1α-mGM-CSF-IRES-hrGFP).
- Generate the GM-CSF cDNA insert.
- Digest the plasmid pMSCV-mGM-CSF-IRES-EGFP19 (20 µg) using the restriction enzyme EcoRI (100 units) at 37 °C for 2 h to produce two DNA fragments: the vector backbone (6.5 kb) and the murine GM-CSF cDNA insert (474 bp).
- Resolve the digested DNA through agarose gel electrophoresis (2%). Purify the murine GM-CSF cDNA fragment (474 bp) using a DNA gel extraction kit.
- Generate the expression plasmids.
- Set up 2 ligation reactions (10 μL total volume) using a DNA ligation kit.
- To generate the empty vector pEF1α-IRES-hrGFP, ligate the untreated vector backbone from step 2.1.3 (6.0 kb; 500 ng).
- To generate pEF1α-mGM-CSF-IRES-hrGFP, ligate the following DNA fragments: (1) the dephosphorylated vector backbone from step 2.1.3 (6.0 kb; 500 ng) and (2) the mGM-CSF cDNA fragment generated in step 2.2.2 (474 bp; 200 ng).
- Ligate at 25 °C for 5 min. Transform ligated DNA into DH5α competent E. coli cells. Plate the transformed E. coli cells on LB-agar plates containing carbenicillin (50 μg/mL).
- Purify plasmids from single E. coli colonies using a DNA isolation kit. Validate the identities of the plasmids by DNA sequencing.
- Set up 2 ligation reactions (10 μL total volume) using a DNA ligation kit.
3. Generation of ES-D3 cells overexpressing GM-CSF
NOTE: Transfect the ES-D3 cells with the plasmid pEF1α-mGM-CSF-IRES-hrGFP to overexpress GM-CS. Cotransfect the plasmid pBabe-Neo into ES-D3 cells to facilitate selection of stably transfected cells18,20.
- Transfect the plasmids into the ES-D3 cells.
- Plate the ES-D3 cells (1.4 x 106) in a gelatin-coated (0.1%) 10 cm tissue culture dish with culture medium (10 mL) for transfection. Culture plated ES-D3 cells at 37 °C for 24 h.
- Prepare two plasmid mixtures in 1.5 mL microcentrifuge tubes: (1) pEF1α-IRES-hrGFP (28 µg, vector control) with pBabe-Neo (4 µg) and (2) pEF1α-mGM-CSF-IRES-hrGFP (28 µg, expressing GM-CSF) with pBabe-Neo (4 µg). Carry out transfection using a transfection kit following the manufacturer's protocol.
- Add transfection medium (1 mL) and transfection reagent (64 µL) to each tube containing the plasmid mixtures and incubate at room temperature for 5 min.
- Add the transfection mixtures to respective 10 cm dishes of ES-D3 cells. Incubate at 37 °C for 5 h.
- Replace the medium in 10 cm dishes with fresh culture medium (10 mL). Incubate at 37 °C for 24 h.
- Generate bulk populations of stably transfected ES-D3 cells.
- Remove the medium from transfected ES-D3 cells. Wash the cells with trypsin (2 mL; 0.05%). Add trypsin (2 mL) and incubate at 37 °C for 5 min. Transfer the cells to a 15 mL centrifuge tube and add 2 mL of fresh culture medium to neutralize trypsin. Centrifuge at 390 x g for 5 min.
- Resuspend the transfected cells in fresh culture medium (10 mL). Evaluate the fluorescence intensity of GFP in transfected ES-D3 cells using fluorescence-activated cell sorting (FACS), following manufacturer's protocol.
- Transfer the transfected cells into two 10 cm dishes containing fresh culture medium (10 mL). Add neomycin (0.5 mg/mL) to eliminate untransfected cells.
- Continue culturing the transfected cells in culture medium containing neomycin (0.5 mg/mL). When the transfected ES-D3 reach 90% confluency, transfer cells to 15 cm tissue culture dishes again. Repeat the procedure for 2 weeks.
- Generate clones of stably transfected ES-D3 cells.
- Once bulk populations of stably transfected ES-D3 cells are generated, collect the cells as before. Determine the cell numbers using a hemocytometer. Centrifuge the cells at 390 x g for 5 min. Resuspend the cells (1 x 107 cells/mL) in fresh culture medium.
- Filter the cells through a sterile 40 µm cell strainer. Purify GFP-positive ES-D3 cells using FACS, following manufacturer's protocol.
- Plate a single sorted ES-D3 cell into one well of a gelatin-coated (0.1%) 96-well tissue culture plate containing parental ES-D3 cells (1 x 103) in neomycin-free medium (200 µL). Co-culturing transfected ES-D3 cells with their untransfected parental counterparts ensures that stably transfected single ES-D3 cells survive and proliferate as a single clone.
- Culture the cells for 48 h, and then add neomycin (0.5 mg/mL) to 96-well plates to eliminate untransfected parental ES-D3 cells.
- Continue culturing the GFP-positive ES-D3 cells in 96-well tissue culture plates with medium containing neomycin (0.5 mg/mL) for 1 week. Transfer clonal ES-D3 cell lines to gelatin-coated (0.1%) 6 cm tissue culture dishes with culture medium (5 mL) containing neomycin (0.5 mg/mL) for 1 week.
- Determine the intensity of GFP fluorescence in each of transfected ES-D3 cell clones using FACS, following manufacturer's protocol. Select the ES-D3 clones expressing either GM-CSF or the empty vector with high levels of green fluorescence.
- Determine the amounts of GM-CSF secreted by ES-D3 cells using a murine GM-CSF ELISA kit, following manufacturer's protocol.
4. Isolation of exosome-enriched extracellular vesicles
- Culture the ES-D3 cells (1 x 107) in 15 cm tissue culture dishes for 72 h at 37 °C. Collect the cell culture supernatant. Store collected supernatant at 4 °C up to 1 week to maintain exosomal integrity.
- Centrifuge the cell culture supernatant at 5,000 x g for 60 min at 4 °C using a centrifuge to sediment large cell fragments.
- Collect the supernatant and centrifuge at 100,000 x g for 90 min at 4 °C using an ultracentrifuge.
- Remove the supernatant. Gently rinse each pellet twice with phosphate-buffered saline (PBS; 1 mL) to remove residual culture supernatant.
- Resuspend each pellet in PBS. Quantify exosome-enriched EVs by their protein content5.
- Measure the protein concentration of exosome-enriched EVs with a bicinchoninic acid (BCA) assay. The expected yield of exosome-enriched EVs from ES-D3 cells is approximately 4 µg protein/mL of cell culture supernatant. Resuspend the exosome-enriched EVs in PBS (protein concentration: ~6 µg/µL). Store the exosome-enriched EVs at -80 °C.
5. Characterization of exosome-enriched extracellular vesicles by transmission electron microscopy
NOTE: Investigate the composition and the structure of the exosome-enriched EVs isolated from ESCs using transmission electron microscopy (TEM)5.
- Fix the exosome-enriched EVs (3-5 µg/µL) with a final concentration of 2% EM grade paraformaldehyde at room temperature for 2 h.
- Load fixed samples (10 µL) onto copper grids with carbon support film. Incubate the samples with copper grids for 1 min, and then drain the grids with filter paper.
- Stain the grids with a staining solution following manufacturer's protocol.
- Transfer the grids to a piece of filter paper using tweezers. Allow the grids to dry overnight at room temperature.
- Acquire electron microscopy images using a transmission electron microscope (50,000x magnification), following manufacturer's protocol.
6. Evaluation of exosome-enriched extracellular vesicles by western blot analysis
- Prepare whole cell extracts.
- Remove the medium from ES-D3 cells cultured in 15 cm dishes. Wash the cells with trypsin (5 mL; 0.05%). Add trypsin (5 mL) to the cells. Incubate at 37 °C for 5 min. Collect the cells and add fresh culture medium (5 mL) to neutralize trypsin. Centrifuge the cells at 390 x g for 5 min. Resuspend the cells in PBS.
- Determine cell numbers using a hemocytometer. Centrifuge the cells again at 390 x g for 5 min. Resuspend the cells in SDS-PAGE loading buffer containing 0.5% SDS (5,000 cells/µL).
- Sonicate the samples for 10 s using a sonicator with 10% amplitude (wattage: 500 W; ultrasonic frequency: 20 kHz). Heat the samples at 100 °C for 5 min.
- Prepare the lysates of exosome-enriched EVs.
- Resuspend the exosome-enriched EVs in SDS-PAGE loading buffer containing 0.5% SDS at a concentration of 1.2 µg/µL.
- Sonicate the samples for 10 s using a sonicator with 10% amplitude (wattage: 500 W; ultrasonic frequency: 20 kHz). Heat the samples at 100 °C for 5 min.
- Detect proteins by Western blot.
- Load whole cell extracts (10 µL; 5,000 cells/µL) and exosome-enriched EV lysates (10 µL; 1.2 µg/µL) into each well of a Bis-Tris PAGE gel (4-20%). Transfer proteins onto polyvinylidene fluoride (PVDF) membranes.
- Incubate membranes with appropriate primary and secondary antibodies. Dilute antibodies (at the concentrations indicated below) in blotting buffer containing PBS, Tween-20 (0.2%), and nonfat dry milk (10% w/v).
- Use the following primary antibodies: anti-Annexin V (200 ng/mL), anti-CD81 (50 ng/mL), anti-Flotillin-1 (200 ng/mL), anti-cytochrome c (100 ng/mL), anti-protein disulfide isomerase (200 ng/mL), anti-GAPDH (33 ng/mL), and anti-Oxphos COX IV-subunit IV (600 ng/mL).
- Use the following secondary antibodies: peroxidase-conjugated goat anti-rabbit IgG (20 ng/mL) and peroxidase-conjugated goat anti-mouse IgG (20 ng/mL).
- Detect proteins using an enhanced chemiluminescence detection kit.
7. Determining GM-CSF concentrations in exosome-enriched extracellular vesicles by ELISA
NOTE: Evaluate the amounts of GM-CSF in exosome-enriched EVs by ELISA using a kit for murine GM-CSF, following manufacturer's protocol with some modifications.
- Coat the ELISA plate with capture antibody. Treat exosome-enriched EVs (0.6 µg) in PBS alone or PBS + 0.05% Tween-20 (100 µL) at room temperature for 30 min. Add treated samples to the coated ELISA plate and incubate at room temperature for 1 h. Wash the plate with PBS alone or PBS + 0.05% Tween-20.
- Add detection antibody to the samples. Incubate at room temperature for 1 h. Wash the plate with PBS alone or PBS + 0.05% Tween-20. Add Avidin-HRP to the samples. Incubate at room temperature for 30 min. Wash the plate with PBS alone or PBS + 0.05% Tween-20.
- Determine the concentrations of GM-CSF in exosome-enriched EVs by measuring the absorbance at 450 nm on a microplate reader.
Wyniki
GM-CSF is overexpressed in murine ESCs.
To stably overexpress GM-CSF in ES-D3 cells, murine GM-CSF cDNA was cloned into a transfection vector to generate the expression vector pEF1α-mGM-CSF-IRES-hrGFP (Figure 1A). GM-CSF was overexpressed in ES-D3 cells by transfection, and about 20% of transiently transfected ES-D3 cells were GFP-positive. Cell clones stably overexpressing GM-CSF or the empty vector control were acquired by FACS. As shown in Figure 1B, the GFP fluorescence intensity of a GM-CSF-expressing ES-D3 cell line or an ES-D3 cell line expressing the empty vector was much higher than that of their parental counterparts. An ELISA assay was carried out to evaluate the GM-CSF concentrations in the cell culture supernatant of different cell lines (Figure 2). ES-D3 cells expressing GM-CSF produced markedly higher levels of GM-CSF in the cell culture supernatant than their empty vector control. Furthermore, the amount of GM-CSF generated by GM-CSF-expressing ES-D3 cells was similar to that of STO fibroblasts expressing GM-CSF, as reported previously19.
Exosomes are enriched in extracellular vesicles derived from murine ESCs.
Vector control and GM-CSF-expressing ES-D3 cell cultures were expanded, and cell culture supernatant was collected. EVs were isolated after several steps of centrifugation. Single EVs were first evaluated by TEM (Figure 3). As shown in the TEM images, isolated EVs contained vesicles of different sizes, which is commonly observed in exosomal preparations5. Importantly, the diameters of the individual vesicles were 30-100 nm, consistent with earlier reports describing exosomes21. Furthermore, the presence of exosomes in EVs was examined by Western blotting (Figure 4). The expression of exosomal markers, including CD81, annexin V, and Flotillin-1, was markedly enhanced in EVs isolated from ES-D3 cells compared with corresponding whole cell extracts (WCE). Importantly, the presence of other subcellular compartment markers in ES-D3-derived EVs was not detected, including (1) the endoplasmic reticulum (ER) marker protein disulfide isomerase (PDI), (2) the mitochondrial markers cytochrome c and COX IV-subunit IV, and (3) the cytosolic marker GAPDH. Overall, these data demonstrate that exosomes were highly enriched in EVs derived from ES-D3 cells.
GM-CSF is localized inside exosome-enriched extracellular vesicles isolated from ESCs.
To determine whether exosome-enriched EVs contain GM-CSF molecules, ELISA assay was conducted to evaluate the levels of GM-CSF in exosome-enriched EVs acquired from ES-D3 cells with or without GM-CSF expression (Figure 5). To further investigate GM-CSF protein localization within exosome-enriched EVs, the GM-CSF levels were quantitated in exosome-enriched EVs under different washing conditions by ELISA. For this purpose, the detergent Tween-20 (0.05%) was first employed to permeabilize the exosomal membranes, and ELISA assays were carried out in the buffers with or without 0.05% Tween-20. Because Tween-20 is known to reduce protein-protein interactions, the background GM-CSF levels detected in the control EVs were significantly reduced by Tween-20 in the washing buffer. In contrast, GM-CSF levels in the EVs of GM-CSF-expressing cells were significantly increased by Tween-20. These results demonstrate that Tween-20-induced exosomal membrane permeabilization makes GM-CSF molecules inside the vesicles accessible for antibody recognition, providing evidence that the majority of exosomal GM-CSF molecules are localized inside the lumen of isolated vesicles.
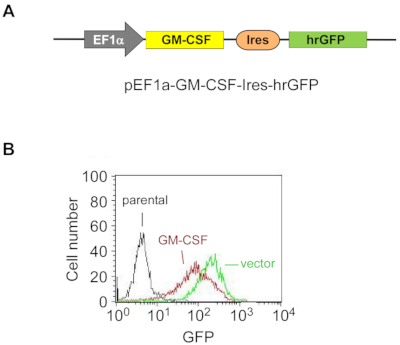
Figure 1: Exogenous GM-CSF is stably overexpressed in ES-D3 cells.
(A) The schematic diagram of the plasmid for overexpressing murine GM-CSF in ES-D3 cells, in which an EF1α promoter drives GM-CSF expression and hrGFP serves as an expression marker.
(B) Fluorescence intensity of GFP in GM-CSF-expressing ES-D3 cells or their empty vector control counterparts was determined by FACS. Please click here to view a larger version of this figure.
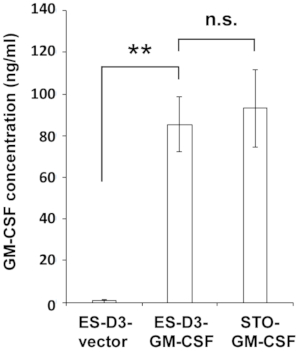
Figure 2: ES-D3 cells overexpressing GM-CSF produce high levels of GM-CSF.
GM-CSF concentrations in the medium of the indicated cells were measured by ELISA. The data are presented as mean ± standard deviations (mean ± SD) of three independent ELISA measurements: **p < 0.001, NS = not significant, ANOVA with Tukey's multiple comparison test. Please click here to view a larger version of this figure.

Figure 3: ES-D3-derived extracellular vesicles are examined by transmission electron microscopy.
Extracellular vesicles were prepared from ES-D3 cells transfected with the plasmid expressing GM-CSF or its empty vector counterpart. Arrows indicate individual vesicles. Scale bar = 100 nm. Please click here to view a larger version of this figure.
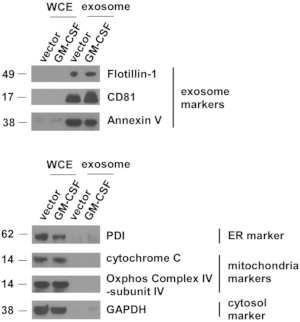
Figure 4: Exosomal markers are highly concentrated in extracellular vesicles isolated from ES-D3 cells.
The amounts of markers for exosomes, endoplasmic reticulum (ER), mitochondria, and cytosol in the indicated whole cell extracts (WCE) and EVs were evaluated by Western blotting. PDI = protein disulfide isomerase. Molecular weights markers (kD) are on the left. Please click here to view a larger version of this figure.

Figure 5: Evaluation of GM-CSF levels in exosome-enriched extracellular vesicles.
The levels of GM-CSF in the indicated exosome-enriched EVs were determined under different ELISA conditions. Exosome-enriched EVs were pretreated with or without 0.05% Tween-20. ELISA was carried out using washing buffer containing either PBS only or PBS + 0.05% Tween-20. The data are presented as the mean ± SD of three independent ELISA assays. *p < 0.05, **p < 0.005, ANOVA with Tukey's multiple comparison test. Please click here to view a larger version of this figure.
Dyskusje
This study shows a highly efficient method of producing exosome-enriched EVs carrying the immune-stimulatory protein GM-CSF, which can be employed to study the immune-modulatory effects of exosome-enriched EVs. Several studies suggest that exosomes exhibit immune-regulatory and anti-tumor functions22. Thus, exosomes from ESCs expressing GM-CSF might also possess biological activities that regulate the immune response. In this protocol, exogenous murine GM-CSF was stably overexpressed in murine ES-D3 cells by transfection (Figure 1). Importantly, significant amounts of GM-CSF were detected in exosome-enriched EVs isolated from ES-D3 cells overexpressing GM-CSF (Figure 5).
These data suggest that almost all GM-CSF resides within exosome-enriched EVs. As a cytokine, the majority of GM-CSF protein is secreted extracellularly11. Like other exosomal cargo material (e.g., mRNAs, miRNAs, and proteins), GM-CSF molecules in the cytosol are encapsulated in the intraluminal vesicles (ILVs) of multivesicular bodies (MVBs)6,23. Upon fusion of MVBs with the plasma membrane, GM-CSF-carrying ILVs are released into the extracellular space.
An important step in this protocol is to effectively overexpress GM-CSF in murine ES-D3 cells (Figure 2). Earlier efforts to achieve this goal by retroviral infection largely failed, likely because of the suppression of retroviral gene expression at the transcriptional levels in ESCs24. Among several viral and cellular promoters examined, the human EF1α promoter showed the most robust activity in ES-D3 cells. Importantly, transgene expression under the control of the EF1α promoter remained stable following long-term cell culture of transfected ES-D3 cells. To express exogenous GM-CSF in another cell type, further studies are needed to evaluate the efficiency of various promoters. The application of this method to isolate GM-CSF-bearing exosome-enriched EVs can be expanded to other stem cell types as well as tumor cells engineered to express various cytokines. Like GM-CSF, most cytokines are secreted extracellularly25. Therefore, a possible limitation of this approach is that the amount of a given cytokine accumulated in exosome-enriched EVs is too low to exhibit its biological activity. For a particular cytokine, new studies need to be carried out to optimize its protein level and biological activity in exosome-enriched EVs.
The most critical hurdle in this study is generating ES-D3 clones overexpressing GM-CSF. To maintain the pluripotential state and promote cell proliferation in vitro, murine ESCs are generally cultured in the presence of feeder cells26. To acquire exosomes exclusively from ESCs, a feeder-cell-free protocol to culture ESCs27 was employed. In this study, ES-D3 cells were cultured in gelatin-coated dishes using medium supplemented with LIF. The plate efficiency and proliferation of single clones of ES-D3 cells under this culture condition were extremely low, making it very challenging to generate ES-D3 clones from single cells. The addition of the conditional medium obtained from parental ES-D3 cells failed to rescue the proliferation deficiency of plated single ES-D3 cells. To overcome this limitation, single ES-D3 cells overexpressing GM-CSF were plated along with parental ES-D3 cells. This plating approach improved the viability of GM-CSF-expressing single ES-D3 clones, facilitating clonal proliferation and expansion. Once transfected, single ES-D3 clones apparently attached to tissue culture plates. They proliferated regardless of the presence of other ES-D3 cells, as parental ES-D3 cells were eliminated 48 h after being plated, allowing only transfected single ES-D3 cells to grow.
Overall, a protocol to successfully generate exosome-enriched EVs carrying GM-CSF from ESCs with the potential to stimulate the immune response in different disease conditions was developed. Furthermore, our recently published study demonstrates that GM-CSF-bearing exosome-enriched EVs from ESCs can serve as a cell-free prophylactic vaccine against cancer28.
Ujawnienia
Kavitha Yaddanapudi, Chi Li, and John W. Eaton submitted a US patent application "Compositions comprising engineered embryonic stem cell-derived exosomes and methods of use thereof."
Podziękowania
We are grateful to Mr. Arkadiusz Slusarczyk and Kentucky Biomedical Research Infrastructure Network (KBRIN, P20GM103436) for acquiring transmission electron microscope images. This work was supported in part by grants from NIH AA018016-01 (J.W.E.), Commonwealth of Kentucky Research Challenge Trust Fund (J.W.E.), NIH CA106599 and CA175003 (C.L.), NIH CA198249 (K.Y.), and Free to Breathe Research Grant (K.Y.).
Materiały
| Name | Company | Catalog Number | Comments |
| Alkaline phosphate, Calf Intestinal | New England Biolabs | M0290S | Dephosphorylating DNA plasmid |
| anti-Annexin V mAb | Santa Cruz Biotechnology | clone H-3, sc-74438 | Western blot, RRID:AB_1118989 |
| anti-CD81 mAb | Santa Cruz Biotechnology | clone B-11, sc-166029 | Western blot, RRID:AB_2275892 |
| anti-cytochrome c mAb | Santa Cruz Biotechnology | clone A-8, sc-13156 | Western blot, RRID:AB_627385 |
| anti-Flotillin-1 mAb | Santa Cruz Biotechnology | clone C-2; sc-74566 | Western blot, RRID:AB_2106563 |
| anti-GAPDH pAb | Rockland | 600-401-A33S | Western blot, RRID:AB_11182910 |
| anti-mouse IgG, goat, peroxidase-conjugated | Thermo Fisher | 31430 | Western blot, RRID:AB_228307 |
| anti-Oxphos COX IV-subunit IV mAb | Thermo Fisher | clone 20E8C12 A21348 | Western blot, RRID:AB_221509 |
| anti-protein disulfide isomerase (PDI) pAb | Enzo | ADI-SPA-890 | Western blot, RRID:AB_10616242 |
| anti-rabbit IgG, goat, peroxidase-conjugated | Thermo Fisher | 31460 | Western blot, RRID:AB_228341 |
| BCA (bicinchoninic acid) assay | Thermo Fisher | 23223 | Determining protein concentrations |
| Bis-Tris PAGE Gel, ExpressPlus, 4-20% | Genscript | M42015 | Western blot |
| Carbenicillin, Disodium Salt | Thermo Fisher | 10177012 | Selecting E. coli colonies |
| Centrifuge, Avanti J-26 XPI | Beckman Coulter | Low speed centrifugation | |
| Centrifuge rotor, JA-10 | Beckman Coulter | 09U1597 | Low speed centrifugation |
| Centrifuge bottle, Nalgene PPCO | Thermo Fisher | 3120-0500PK | Low speed centrifugation |
| Cu grids with carbon support film | Electron Microscopy Sciences | FF200-Cu | Acquiring electron microscopy images |
| EcoRI | New England Biolabs | R0101 | Digesting DNA plasmid |
| Enhanced chemiluminescence detection system | Thermo Fisher | 32106 | Western blot |
| FACScalibur flow cytometer | Becton Dickinson | Examining GFP levels of ES-D3 cells | |
| Fetal bovine serum | ATCC | SCRR-30-2020 | Medium for ES-D3 cells |
| Fisherbrand Sterile Cell Strainers; Mesh Size: 40μm | Thermo Fisher | 22-363-547 | Filtering ES-D3 cells for FACS sorting |
| Gelatin (0.1%) | Thermo Fisher | ES006B | Culturing ES-D3 cells |
| GM-CSF ELISA kit | Thermo Fisher | 88733422 | Determining GM-CSF concentrations |
| KnockOut Dulbecco’s Modified Eagle’s Medium | Thermo Fisher | 10-829-018 | Medium for ES-D3 cells |
| Leukemia Inhibitory Factor | Thermo Fisher | ESG1106 | Medium for ES-D3 cells |
| L-glutamine | VWR | VWRL0131-0100 | Medium for ES-D3 cells |
| Lipofectamine 2000 transfection reagent | Thermo Fisher | 11668019 | Transfecting ES-D3 cells |
| Microplate reader, PowerWave XS | BioTek | Determining GM-CSF concentrations | |
| MoFlo XDP high-speed cell sorter | Beckman Coulter | Isolating single ES-D3 cell clones | |
| NEB 5-alpha Competent E. coli | New England Biolabs | C2988J | Generating GM-CSF expression plasmid |
| Neomycin | Thermo Fisher | 10-131-035 | Selecting ES-D3 clones |
| Non-essential amino acids | Thermo Fisher | SH3023801 | Medium for ES-D3 cells |
| Non-fat dry milk | Thermo Fisher | NC9022655 | Western blot |
| Opti-MEM I Reduced Serum Medium | Thermo Fisher | 31985062 | Transfecting ES-D3 cells |
| Paraformaldehyde | Electron Microscopy Sciences | 15710 | Acquiring electron microscopy images |
| Penicillin/streptomycin | VWR | sc45000-652 | Medium for ES-D3 cells |
| Plasmid pEF1a-FD3ER-IRES-hrGFP | Addgene | 37270 | Generating GM-CSF expression plasmid |
| PVDF membranes | Millipore EMD | IPVH00010 | Western blot |
| QIAprep Spin Miniprep Kit (250) | QIAGEN | 27106 | Generating GM-CSF expression plasmid |
| QIAquick Gel Extraction Kit (50) | QIAGEN | 28704 | Generating GM-CSF expression plasmid |
| Quick Ligation Kit | New England Biolabs | M2200S | Generating GM-CSF expression plasmid |
| Transmission electron microscope | Hitachi | HT7700 | Acquiring electron microscopy images |
| Trypsin | VWR | 45000-660 | Culturing ES-D3 cells |
| Ultracentrifuge, OptimaTM L-100 XP | Beckman Coulter | High speed centrifugation | |
| Ultracentrifuge rotor, 45Ti | Beckman Coulter | 09U4454 | High speed centrifugation |
| Ultracentrifuge polycarbonate bottle | Beckman Coulter | 355622 | High speed centrifugation |
| UranyLess staining solution | Electron Microscopy Sciences | 22409 | Acquiring electron microscopy images |
Odniesienia
- Thomson, J. A., et al. Embryonic stem cell lines derived from human blastocysts. Science. 282 (5391), 1145-1147 (1998).
- Sakthiswary, R., Raymond, A. A. Stem cell therapy in neurodegenerative diseases: From principles to practice. Neural Regeneration Research. 7 (23), 1822-1831 (2012).
- Liu, Y. W., et al. Human embryonic stem cell-derived cardiomyocytes restore function in infarcted hearts of non-human primates. Nature Biotechnology. 36 (7), 597-605 (2018).
- Aguayo-Mazzucato, C., Bonner-Weir, S. Stem cell therapy for type 1 diabetes mellitus. Nature Reviews: Endocrinology. 6 (3), 139-148 (2010).
- Thery, C., et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. Journal of Extracell Vesicles. 7 (1), 1535750 (2018).
- Raposo, G., Stoorvogel, W. Extracellular vesicles: exosomes, microvesicles, and friends. Journal of Cell Biology. 200 (4), 373-383 (2013).
- Meldolesi, J. Exosomes and Ectosomes in Intercellular Communication. Current Biology. 28 (8), R435-R444 (2018).
- Stremersch, S., De Smedt, S. C., Raemdonck, K. Therapeutic and diagnostic applications of extracellular vesicles. Journal of Control Release. 244 (Pt B), 167-183 (2016).
- Lindenbergh, M. F. S., Stoorvogel, W. Antigen Presentation by Extracellular Vesicles from Professional Antigen-Presenting Cells. Annual Review of Immunology. 36, 435-459 (2018).
- Kunigelis, K. E., Graner, M. W. The Dichotomy of Tumor Exosomes (TEX) in Cancer Immunity: Is It All in the ConTEXt?. Vaccines (Basel). 3 (4), 1019-1051 (2015).
- Becher, B., Tugues, S., Greter, M. GM-CSF: From Growth Factor to Central Mediator of Tissue Inflammation. Immunity. 45 (5), 963-973 (2016).
- Conti, L., Gessani, S. GM-CSF in the generation of dendritic cells from human blood monocyte precursors: recent advances. Immunobiology. 213 (9-10), 859-870 (2008).
- Higano, C. S., et al. Integrated data from 2 randomized, double-blind, placebo-controlled, phase 3 trials of active cellular immunotherapy with sipuleucel-T in advanced prostate cancer. Cancer. 115 (16), 3670-3679 (2009).
- Yan, W. L., Shen, K. Y., Tien, C. Y., Chen, Y. A., Liu, S. J. Recent progress in GM-CSF-based cancer immunotherapy. Immunotherapy. 9 (4), 347-360 (2017).
- Dranoff, G., et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proceedings of the National Academy of Sciences of the United States of America. 90 (8), 3539-3543 (1993).
- Tremml, G., Singer, M., Malavarca, R. Chapter 1, Unit 1C 4, Culture of mouse embryonic stem cells. Current Protocols in Stem Cell Biology. , (2008).
- Kirsch, P., Hafner, M., Zentgraf, H., Schilling, L. Time course of fluorescence intensity and protein expression in HeLa cells stably transfected with hrGFP. Molecules and Cells. 15 (3), 341-348 (2003).
- Zeng, X., et al. Stable expression of hrGFP by mouse embryonic stem cells: promoter activity in the undifferentiated state and during dopaminergic neural differentiation. Stem Cells. 21 (6), 647-653 (2003).
- Yaddanapudi, K., et al. Vaccination with embryonic stem cells protects against lung cancer: is a broad-spectrum prophylactic vaccine against cancer possible?. PLoS One. 7 (7), e42289 (2012).
- Dalby, B., et al. Advanced transfection with Lipofectamine 2000 reagent: primary neurons, siRNA, and high-throughput applications. Methods. 33 (2), 95-103 (2004).
- Thery, C., Amigorena, S., Raposo, G., Clayton, A. Chapter 3, Unit 3 22, Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Current Protocols in Cell Biology. , (2006).
- Zhang, X., et al. Exosomes for Immunoregulation and Therapeutic Intervention in Cancer. Journal of Cancer. 7 (9), 1081-1087 (2016).
- Bunggulawa, E. J., et al. Recent advancements in the use of exosomes as drug delivery systems. Journal of Nanobiotechnology. 16 (1), 81 (2018).
- Schlesinger, S., Lee, A. H., Wang, G. Z., Green, L., Goff, S. P. Proviral silencing in embryonic cells is regulated by Yin Yang 1. Cell Reports. 4 (1), 50-58 (2013).
- Dranoff, G. GM-CSF-based cancer vaccines. Immunological Reviews. 188, 147-154 (2002).
- Park, Y. G., et al. Effects of Feeder Cell Types on Culture of Mouse Embryonic Stem Cell In Vitro. Development and Reproduction. 19 (3), 119-126 (2015).
- Lin, S., Talbot, P. Methods for culturing mouse and human embryonic stem cells. Methods in Molecular Biology. 690, 31-56 (2011).
- Yaddanapudi, K., et al. Exosomes from GM-CSF expressing embryonic stem cells are an effective prophylactic vaccine for cancer prevention. OncoImmunology. 8 (3), 1561119 (2019).
Przedruki i uprawnienia
Zapytaj o uprawnienia na użycie tekstu lub obrazów z tego artykułu JoVE
Zapytaj o uprawnieniaThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. Wszelkie prawa zastrzeżone