Method Article
Measuring the Motor Aspect of Cancer-Related Fatigue using a Handheld Dynamometer
W tym Artykule
Podsumowanie
Simple and accessible methods were developed to measure the motor aspect of cancer-related fatigue objectively and quantitatively. We describe, in detail, ways to administer the physical fatigue test using a simple handgrip device as well as methods to calculate fatigue indices.
Streszczenie
Cancer-related fatigue (CRF) is commonly reported by patients both during and after receiving treatment for cancer. Current CRF diagnoses rely on self-report questionnaires which are subject to report and recall biases. Objective measurements using a handheld dynamometer, or handgrip device, have been shown in recent studies to correlate significantly with subjective self-reported fatigue scores. However, variations of both the handgrip fatigue test and fatigue index calculations exist in the literature. The lack of standardized methods limits the utilization of the handgrip fatigue test in the clinical and research settings. In this study, we provide detailed methods for administering the physical fatigue test and calculating the fatigue index. These methods should supplement existing self-reported fatigue questionnaires and help clinicians assess fatigue symptom severity in an objective and quantitative manner.
Wprowadzenie
Cancer-related fatigue (CRF) is an prevalent and debilitating symptom that is reported by up to 80% of cancer patients1. The National Comprehensive Cancer Network (NCCN) defines CRF as a persistent sense of physical, emotional, and cognitive exhaustion1. The main differentiating characteristics of CRF are the disproportionality to recent activity and the inability of CRF to be relieved by rest1. As a result, CRF severely impacts patients' participation in daily activities and their health-related quality of life1.
The current assessment of CRF relies primarily on self-report questionnaires2. As a result, symptom severity which is measured using self-reports is subject to recall and reporting biases and can be influenced by the specific questionnaire and cutoff scores used to assess CRF3. As a multidimensional construct, the physical dimension of CRF has been shown to correlate with daily activity changes and a need for daytime naps4, whereas the influence of CRF on physical functioning is less explored. To this date, CRF remains an underdiagnosed and undertreated symptom with no well-defined underlying mechanism or treatment option1. To better understand this debilitating condition, there is an increasing need to measure CRF and its dimensions objectively and quantitatively.
Physical fatigue refers to an inability to maintain the required force during sustained contractile activity5. The subsequent compromised daily functioning as a result of not being able to carry out daily tasks (e.g., carrying grocery bags, lifting and holding an object) greatly affects the health-related quality of life, especially in older adults, and contributes to future injuries6,7. Various tools have been developed to quantify physical impairment including physical performance tests, such as the 6 min walk test (6MWT) and sit-to-stand test (STS), as well as wearable physical activity monitors, such as actigraphy devices and fitness trackers8,9,10. Physical performance tests such as 6MWT and STS are easy to administer and do not require special equipment10. However, the reliability and success of such tests require clinician training and logistical requirements such as a 30 m corridor10. Wearable activity monitors allow for automated data collection and longitudinal symptom monitoring11. However, these activity monitors often need to be worn for multiple days, and patient compliance can be an issue11. In addition, the large amount of data collected using activity monitors can be challenging to process, making it difficult to derive clinically meaningful information11.
The handheld dynamometer, or instrumented handgrip device with computer-assisted data acquisition, is a portable apparatus that measures grip strength. Handheld dynamometry has been used to test motor fatigue and impairment in disease conditions that typically involve the motor system including motor neurons and muscular problems12. Recent work has demonstrated an association between self-reported subjective CRF scores and motor fatigue measured using a handgrip static fatigue test13. Handgrip fatigue tests are particularly suitable for clinical use due to their reliability and time efficiency, requiring a few minutes to complete14,15. Furthermore, handgrip fatigue tests can be pre-programed, ensuring data reproducibility7. Administering the handgrip test requires minimal training on the part of the test administrator and can be easily implemented in a clinical setting given a standardized protocol. Using self-reported fatigue questionnaires in conjunction with the handgrip fatigue test should provide additional tools for clinicians to screen, monitor, and manage fatigue symptoms in cancer patients.
The lack of standardized consensus methods has limited the adoption of the handgrip fatigue test in the clinics16. In this current work, we outline three different methods to use the handheld dynamometer to quantify motor fatigue objectively. The utility of each method should be tested in each cancer population to ensure it accurately distinguishes between fatigued and non-fatigued subjects. We also outline methods to calculate the fatigue index for each handgrip fatigue test. The goal of this work is to provide a comprehensive toolkit to supplement self-reported questionnaires and to standardize CRF physical performance measurement accurately and objectively.
Protokół
The current study (NCT00852111) was approved by the Institutional Review Board (IRB) of the National Institutes of Health (NIH). Participants enrolled in this study were 18 years of age or older, diagnosed with non-metastatic prostate cancer with or without prior prostatectomy, and scheduled to receive external beam radiation therapy at the Radiation Oncology Clinic of the NIH Clinical Center. Potential participants were excluded if they had a progressive disease that could cause significant fatigue, had psychiatric disease within the past five years, had uncorrected hypothyroidism or anemia, or had a second malignancy. Individuals who used sedatives, steroids, or non-steroidal anti-inflammatory agents were also excluded. All participants were recruited at the Magnuson Clinical Research Center at the NIH. Signed written informed consents were obtained prior to study participation.
1. Handgrip preparation and testing position
- In a quiet room, set up a chair with armrests.
- Turn on the handheld dynamometer.
- The software will prompt calibration of the dynamometer. Ensure the device is resting on a flat surface during calibration.
- Seat the subject in an upright position with their feet in full contact with the floor and hips as far back as the chair supports.
- Ensure the subject's hip and knee angles are close to 90° and shoulders are in neutral abduction/adduction and neutrally rotated. Ensure the subject's elbow is flexed at 90° and the wrist is unsupported, as recommended by the American Society of Hand Therapists handbook17.
- After calibrating the dynamometer, instruct the subject to grasp the dynamometer, with the dorsal intermediate phalanges facing forward.
- Adjust the grip position to the subject's hand size and record it7.
- Maintain the same handgrip testing position for all subsequent tests.
- Prior to each test, provide standardized scripts and ask subjects to perform a mock attempt to demonstrate understanding of the instructions.
- Inform the subjects that discomfort is normal, but the tests can be discontinued in the presence of unexpectedly severe strain/pain.
- Stop the test if severe discomfort is reported by the patient or in the event of unexpected circumstances.
- Ensure a 2 min rest period between trials to allow the muscle to recover18.
2. Maximal voluntary isometric contraction (MVIC) test
- Provide subjects with standardized instructions. For example, "in the test, you will squeeze as hard as you can for 5 s, starting with your non-dominant hand. This test will be done three times for each hand. For each test, I will count down 3, 2, 1...GO. Squeeze the device on GO as hard as you can."
- On "Go", start the program by clicking on the GO button.
- Repeat the MVIC test for a total of three times with a 30 s rest between trials.
- The average for each hand from the three trials maximum force is the MVIC19.
3. Maximum force static fatigue test
- Instruct subjects to exert full effort to achieve maximal contraction during the static fatigue test.
- On "Go", start the program by clicking on the GO button. Use standardized encouragement script such as squeeze hard repeatedly until the test ends.
- Continue the static fatigue test for 35 s, so as to provide up to 5 s to achieve Fmax (maximal handgrip strength).
- Static fatigue index (SFI)12,20,21
- Calculate SFI using the following equation:
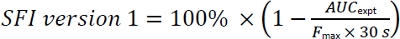
- Calculate AUCexpt by computing the experimental area under the curve from the time that Fmax was achieved (Tmax) to 30 s after Tmax.
- Calculate the hypothetical AUC (AUChypothetical) in the absence of fatigue by multiplying the Fmax by 30 s.
NOTE: Higher SFI values indicate increased divergence from the expected value, hence higher fatigue. - Calculate SFI version 2 as the ratio of the maximal force during the last 5 s (Fmax 25-30s) to the maximal force in the first 5 seconds (Fmax 0-5s) using the equation:

NOTE: Higher values of SFI indicate higher fatigue.
- Calculate SFI using the following equation:
4. Sub-maximum force static fatigue test
- Indicate the value of 50% of the MVIC of the participant's non-dominant hand by drawing a horizontal line on a transparency overlay of the screen.
- Draw a second line on the overlay in a different color to indicate a 10% decline of the target value.
- Ensure the participant can easily see the screen and 50% MVIC line.
- Instruct the subject to maintain a target value of 50% of MVIC for as long as possible.
- Count down. On "Go", start the program by clicking on the GO button.
- Stop the test when the strength declines by 10% of the target value for more than 5 s as indicated by the second line on the transparency.
- Calculate total work performed7 as the force-versus-time area under the curve over the period of time during which the target force (T50% MVIC) is sustained:
Total work = AUC during T50% MVIC
NOTE: Endurance can be measured as time to task completion22. Higher values of total work indicate lower fatigue.
5. Dynamic fatigue test
- Instruct subjects to perform a maximal squeeze every second for a duration of 30 s. Use a metronome to provide rhythm guidance20.
- Start the metronome which is set at 1 beep per second.
- Start the countdown. On "Go", start the test by clicking on the GO button. Ensure the countdown matches the rate of the metronome.
- Inform the participant when passing the halfway point and when 5 s are remaining.
- Stop test after the 30 s are completed.
- Dynamic Fatigue Index
- Calculate the Dynamic Fatigue Index20 using the maximal force (Fmax) of the last 5 s and the Fmax of the first 5 s.

NOTE: Higher values of the dynamic fatigue index (DFI) indicate higher fatigue.
- Calculate the Dynamic Fatigue Index20 using the maximal force (Fmax) of the last 5 s and the Fmax of the first 5 s.
Wyniki
Representative force (kg) versus time (s) traces are shown in Figure 1. During the static fatigue test, subjects typically reach maximal strength (Fmax) within 2–3 s23. Self-reported fatigue in subjects was measured based on previous studies3. The absence of Fmax (±10% MVIC) within 3 s indicates insufficient effort23. To prevent this issue, verbal encouragement should be provided. Both subjects reporting fatigue (black line) and no fatigue (gray line) reached Fmax within 5 s, and force gradually declined over the course of the static fatigue test (Figure 1A). During the submaximal fatigue test, subjects are instructed to reach and maintain 50% of the previously determined MVIC and are provided with visual guidance during the test. Once 50% MVIC was reached, both non-fatigued and fatigued subjects maintained steady force output for an extended period of time (Figure 1B). For the dynamic fatigue test, subjects were instructed to exert maximal force at 1 contraction/s. Both the non-fatigued and fatigued subjects maintained steady output until the end of the test (Figure 1C). Subjects typically report the highest levels of difficulty during the static fatigue test, whereas both the submaximal fatigue test and the dynamic fatigue test were well-tolerated.
Fatigue index calculations are illustrated in Figure 2. Static fatigue index (SFI) version 1 (Figure 2A) represents the difference between the actual force generated (AUCexpt) and the hypothetical condition in the absence of fatigue (Fmax multiplied by 30 s). As different subjects reach Fmax at different times, this method finds the time when Fmax is achieved (Tmax) and only takes into account the force generated from Tmax to 30s afterwards. An alternative static fatigue index calculation is shown in Figure 2B. This method represents the decline in force from the first 5 s of the test (Fmax 0-5s) to the last 5 s of the test (Fmax 25-30s). Higher values of both static fatigue indices represent higher levels of fatigue. Performance on the submaximal fatigue test is evaluated using total work, which is calculated as the cumulative force generated (AUC) during the target range at 50% MVIC (Figure 2C). Higher values of total work represent lower fatigue. The dynamic fatigue index represents the decline in intermittent contractile force from the first 5 s (Fmax 0-5s) to the last 5 s (Fmax 25-30s) (Figure 2D). Higher values of the dynamic fatigue index represent higher levels of fatigue.
Using the same static fatigue test force-versus-time traces (shown in Figure 1), we found that static fatigue index calculation version 1 resulted in a better dissociation between non-fatigued (SFI = 26.65%) and fatigued (SFI = 29.14%) subjects (Figure 3A). In contrast, while static fatigue index version 2 also detected differences between non-fatigued (SFI = 33.56%) and fatigued (SFI = 35.02%) subjects, the difference between the two groups was smaller compared to static fatigue index version 1 (Figure 3B). Using the submaximal fatigue test, the non-fatigued subject exhibited higher endurance (69.75 s) and total work performed at a target level of 50% MVIC (1,244.45 kg·s) compared to the fatigued subject both in terms of endurance (67.36 s) and total work performed (931.252 kg·s) (Figure 3C). The dynamic fatigue index also captured the difference between non-fatigued (SFI = 10.94%) and fatigued (SFI = 13.84%) (Figure 3D). However, we have observed differences in subjects' ability to adhere to a consistent rhythm even when guided with a metronome, which introduces variability in the total force exerted during each intermittent contraction.

Figure 1: Sample force-time traces. Representative traces of the (A) static fatigue test, (B) submaximal fatigue test, and (C) dynamic fatigue test are plotted as force (kg) versus time (s) graphs. Non-fatigue subject traces are shown in gray, fatigue subject traces are shown in black. Please click here to view a larger version of this figure.
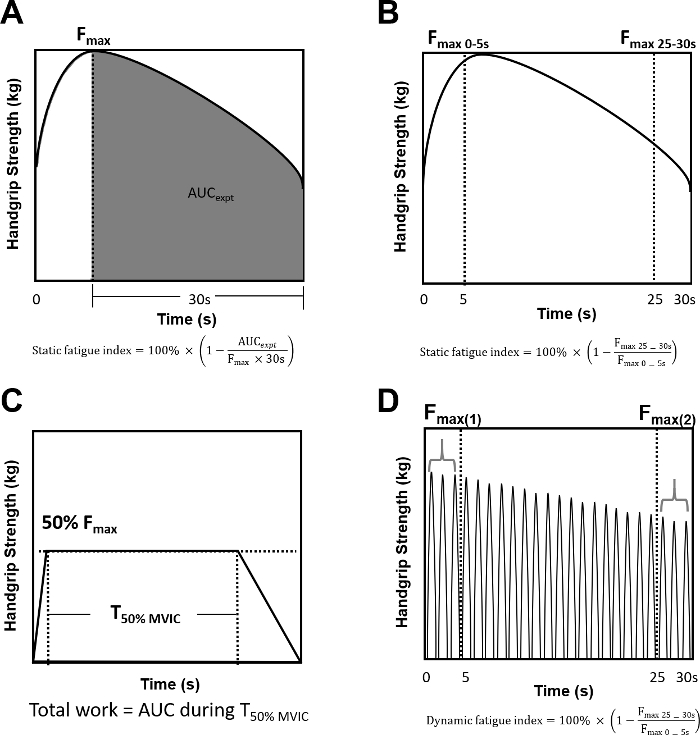
Figure 2: Illustrations of fatigue index calculations. (A) Static fatigue index calculation version 1. (B) Static fatigue index calculation version 2. (C) Submaximal fatigue test total work calculation. (D) Dynamic fatigue index calculation. Please click here to view a larger version of this figure.

Figure 3: Representative data collected using methods outlined in the protocol. (A) Static fatigue index calculation version 1. (B) Static fatigue index calculation version 2. (C) Submaximal fatigue test total work calculation. (D) Dynamic fatigue index calculation. Please click here to view a larger version of this figure.
Dyskusje
Here, we provide three different methods for measuring the physical dimension of CRF. Motor fatigue tests using handheld dynamometers are simple and easily adaptable for clinical use. Since many variations of the test exist in the literature, our goal was to provide standardized methods to administer these tests and decrease the need for extensive in-person trainings for clinicians.
Although the fatigue tests outlined in this study demonstrate good test-retest reliability7,20, adherence to this protocol will ensure data reproducibility. A critical and often overlooked step during test preparation is to allow the handgrip device to calibrate on a flat surface. This step will establish an actual baseline reading. Even though maximal strength is not an accurate indicator for the physical aspect of CRF13, obtaining the true MVIC value will greatly enhance data interpretation. It is used to determine if motor weakness is present via normative data comparisons24. Accurate MVIC values also ensure that the static fatigue test is an actual maximal performance test, facilitating normative comparisons for fatigue indices. Motor fatigue tests are particularly helpful in a clinical setting where the static fatigue test may be used as a screening tool in addition to making longitudinal comparisons. We recommend that Fmax be inspected and be within 10% of MVIC to ensure the maximal static fatigue test does not become a de facto submaximal test. Consistent with previous studies, we found that verbal encouragement during handgrip fatigue tests is necessary for achieving reproducible data and a good Fmax (± 10% of MVIC)25,26. Maintaining maximal contraction, particularly during the static fatigue test, requires concentration and motivation. In the absence of verbal encouragement, subjects at times fail to achieve the true maximal contraction during the first 5 s or during the entire test, which introduces variability in static fatigue index (SFI) calculations. Related to this point, a standard script should be used when providing instructions before the test and during verbal encouragement, consistent with previous studies25,26.
SFI version 1 (Figure 2A) represents the difference between curves of the actual force-versus-time and the hypothetical force-versus-time in the absence of fatigue. Multiple variations of the calculations have been developed in previous studies20. Since subjects typically reach maximal force within the first 5 s, the following modifications can be used to calculate the actual area under the curve (AUCexpt): (1) AUC from 5 to 30 s of the test, (2) the entire duration of the test from 0–30 s, and (3) the total AUC from when Fmax is achieved (Tmax) to 30 s after that12,20. The time interval used to calculate AUCexpt value is then used to determine the time interval for calculating the hypothetical AUC (AUChypothetical) in the absence of fatigue, which is typically calculated as Fmax multiplied by the time duration used to derive AUCexpt. In our experience, CRF correlates significantly with the SFI calculated using the method outlined in Figure 2A13. Given the variability of the amount of time necessary to reach Fmax particularly in older patients with cancer, SFI version 1 described in the methods section provides the most sensitive measure to capture the physical dimension of CRF with no loss of data showing early fatiguing, as would occur with looking only at AUC from 5–30 s13.
SFI calculation version 2 (Figure 2B) represents the decline in maximal force generated from the beginning of the test to the end of the test. The calculation method is much simpler than SFI version 1 and provides a quick way to estimate the level of fatigue. However, static fatigue index calculation version 2 exhibited low test-retest reliability with an interclass correlation coefficient (ICC) of 0.46–0.77, while static fatigue index version 1 demonstrated higher test-retest reliability of 0.71–0.96 in Multiple Sclerosis (MS) patients21. This is consistent with our finding that only static fatigue test and SFI calculation version 1 correlated significantly with self-reported fatigue in patients with prostate cancer13. Interestingly, SFI version 1 exhibited lower test-retest reliability (ICC of 0.18-0.52) in healthy controls compared to their MS counterparts21. Therefore, we recommend using SFI calculation version 1 for measuring physical fatigue in patients with cancer. SFI version 2 may be used for providing a quick estimate of fatigue levels during the test.
The submaximal fatigue test consists of either sustained (static) or repetitive (dynamic) contractions at a target value of 30–75% of MVIC. While this test is not typically used to calculate motor fatigability, we included this method in the current protocol because it is often used to induce fatigue during concurrent assessments such as drug treatment effects, blood biomarker analysis, electromyography, and functional magnetic resonance imaging (fMRI)27,28,29. Submaximal fatigue tests cause less discomfort in research subjects30. These tests also better approximate typical daily tasks such as grasping and carrying groceries. In addition, the submaximal fatigue test may be more sensitive to additional treatment-induced effects31. Performance on the submaximal fatigue test can be gauged as total work performed (Figure 2C), which has demonstrated good test-retest reproducibility7. Alternatively, time to task failure, or endurance, has also been used to quantify submaximal fatigue test performance22.
Dynamic fatigue tests consist of intermittent repeated maximal contractions, usually at a fixed rhythm guided with a metronome7. The dynamic fatigue index (DFI) is calculated as the drop in maximal force from the beginning to the end of the test (Figure 2D). The DFI calculation described in the methods considers the maximal force (Fmax) generated during the first and last 3 contractions. This method can be modified to take into account the Fmax in the first 5 s (0–5s) and the last 5 s (25–30 s)20. Similar to the submaximal fatigue test, the dynamic fatigue test causes less discomfort and has good test-retest reliablity7. However, the dynamic fatigue test did not demonstrate sufficient discriminative power in studies comparing fatigued subjects (e.g., post-polio, multiple sclerosis) and healthy controls20.
Motor fatigue measured using the handgrip device may originate from either decreased drive from the descending efferent motor neurons, or impaired contractile mechanisms within muscle fibers5. Interestingly, submaximal contractions may be more influenced by the motor cortex, whereas maximal fatigue tests may have a greater muscular component32. This may be related to the prolonged ischemia induced by high intramuscular pressure (>50% MVIC) and decreased blood flow into muscle tissues29,33. Contractile forces measured using handheld dynamometers involve the adductor pollicis containing predominant Type 1 fibers, as well as forearm flexor digitorium muscles comprised of both Type I and II fibers34. Due to the highly oxidative nature of Type I fibers, maximal static fatigue tests are more prone to muscle ischemia and glycolysis-induced fatigue35. Since submaximal fatigue tests and dynamic/intermittent tests allow for muscle fiber recovery, they may be more useful for assessing physical fatigue with a central origin33. Future studies that aim to isolate contributions of motor neurons versus muscular components may also include transcranial magnetic stimulation of the motor cortex used in conjunction with electromyogram recordings36.
A limitation of the current protocol is that the lack of a well-established normative value for the cancer population using these physical fatigue tests. Previous studies have measured physical fatigue using the handgrip device in populations prone to developing physical fatigue such as polio and aging20,23. Such normative values have yet to be established in cancer fatigue research using standardized methods. In addition, variables such as hand size, the type of handheld dynamometer, the presence of non-slip gloves or rings, hand dominance, gender, age, and baseline fitness can affect handgrip tests. The inevitable heterogeneity of clinical populations may limit the generalizability of study results from using the handgrip test. Therefore, strategies to control for these potential confounding variables should be considered, such as analysis of covariance or MVIC data normalization to body weight. Further, the handgrip test only captures fatiguability of upper limb muscle tissues, which may not correlate with lower limb fatigability26. Careful data interpretation and avoiding over-generalization are, therefore, warranted when using the handgrip test to measure the physical dimension of CRF. It may be helpful to include additional performance fatigability tests that involve lower extremities, such as the 6 min or 10 m walk test, in conjunction with handgrip fatigability tests37. Finally, the methods described in the current study represent motor fatigue measured at a single time point. Previous studies have shown that fatigability, which reflects the change in fatigue during an activity, may be more useful clinically as this concept captures the functional status of the patient38. Future studies will explore the association between perceived fatigability (change in self-reported fatigue scores) and performance fatigability (change in handgrip fatigue indices) before and after physical/cognitive tests37,38,39.
In conclusion, the methods outlined in this protocol provide objective and quantitative measures of a debilitating symptom and are easily applicable in the clinical setting. In our experience, the static fatigue test combined with SFI calculation version 1 is the most sensitive method for capturing the physical aspect of fatigue in cancer as well as other disease conditions12,13. In addition to the static maximal fatigue test, we provided two additional handgrip tests which are less fatiguing and may be better tolerated in severely impaired patient populations. Variables such as age, gender, disease, and baseline fitness level can all affect physical fatigue measurements using the handgrip device. The specific method used should be tailored to each disease population.
Ujawnienia
The authors have nothing to disclose.
Podziękowania
This study is fully supported by the Division of Intramural Research of the National Institute of Nursing Research of the NIH, Bethesda, Maryland.
Materiały
| Name | Company | Catalog Number | Comments |
| Quantitative Muscle Assessment application (QMA) | Aeverl Medical | QMA 4.6 | Data acquisition software. NOTE: other brands/models can be used as long as the software records force over time. |
| QMA distribution box | Aeverl Medical | DSTBX | Software distribution box which connects the handgrip to the software. |
| Baseline hand dynamometer with analog output | Aeverl Medical | BHG | Instrumented handgrip device with computer assisted data acquisition. NOTE: other brands/models can be used as long as the instrument measures force over time |
Odniesienia
- Berger, A. M., et al. Cancer-Related Fatigue, Version 2.2015. Journal of the National Comprehensive Cancer Network : JNCCN. 13 (8), 1012-1039 (2015).
- Campos, M. P. O., Hassan, B. J., Riechelmann, R., Del Giglio, A. Cancer-related fatigue: a practical review. Annals of Oncology. 22 (6), 1273-1279 (2011).
- Feng, L. R., Dickinson, K., Kline, N., Saligan, L. N. Different phenotyping approaches lead to dissimilar biologic profiles in men with chronic fatigue following radiation therapy. Journal of Pain and Symptom Management. 52 (6), 832-840 (2016).
- Minton, O., Stone, P. C. A comparison of cognitive function, sleep and activity levels in disease-free breast cancer patients with or without cancer-related fatigue syndrome. BMJ Supportive & Palliative Care. 2, 231-238 (2012).
- Wan, J. J., Qin, Z., Wang, P. Y., Sun, Y., Liu, X. Muscle fatigue: general understanding and treatment. Experimental & Molecular Medicine. 49 (10), 384 (2017).
- Bautmans, I., Gorus, E., Njemini, R., Mets, T. Handgrip performance in relation to self-perceived fatigue, physical functioning and circulating IL-6 in elderly persons without inflammation. BMC geriatrics. 7, 5-5 (2007).
- Gerodimos, V., Karatrantou, K., Psychou, D., Vasilopoulou, T., Zafeiridis, A. Static and Dynamic Handgrip Strength Endurance: Test-Retest Reproducibility. The Journal of Hand Surgery. 42 (3), 175-184 (2017).
- van der Werf, S. P., Prins, J. B., Vercoulen, J. H. M. M., van der Meer, J. W. M., Bleijenberg, G. Identifying physical activity patterns in chronic fatigue syndrome using actigraphic assessment. Journal of Psychosomatic Research. 49 (5), 373-379 (2000).
- Connaughton, J., Patman, S., Pardoe, C. Are there associations among physical activity, fatigue, sleep quality and pain in people with mental illness? A pilot study. Journal of Psychiatric and Mental Health Nursing. 21 (8), 738-745 (2014).
- Gurses, H. N., Zeren, M., Denizoglu Kulli, H., Durgut, E. The relationship of sit-to-stand tests with 6-minute walk test in healthy young adults. Medicine. 97 (1), 9489 (2018).
- Beg, M. S., Gupta, A., Stewart, T., Rethorst, C. D. Promise of Wearable Physical Activity Monitors in Oncology Practice. Journal of Oncology Practice. 13 (2), 82-89 (2017).
- Severijns, D., Lamers, I., Kerkhofs, L., Feys, P. Hand grip fatigability in persons with multiple sclerosis according to hand dominance and disease progression. Journal of Rehabilitation Medicine. 47 (2), 154-160 (2015).
- Feng, L. R., et al. Cognitive and motor aspects of cancer-related fatigue. Cancer Medicine. 8 (13), 5840-5849 (2019).
- Bohannon, R. W. Hand-Grip Dynamometry Predicts Future Outcomes in Aging Adults. Journal of Geriatric Physical Therapy. 31 (1), 3-10 (2008).
- Reuter, S. E., Massy-Westropp, N., Evans, A. M. Reliability and validity of indices of hand-grip strength and endurance. Australian Occupational Therapy Journal. 58 (2), 82-87 (2011).
- Roberts, H. C., et al. A review of the measurement of grip strength in clinical and epidemiological studies: towards a standardised approach. Age and Ageing. 40 (4), 423-429 (2011).
- American Society of Hand Therapists. . Clinical Assessment Recommendations. 2nd edn. , (1992).
- Bhuanantanondh, P., Nanta, P., Mekhora, K. Determining Sincerity of Effort Based on Grip Strength Test in Three Wrist Positions. Safety and Health at Work. 9 (1), 59-62 (2018).
- van Meeteren, J., van Rijn, R. M., Selles, R. W., Roebroeck, M. E., Stam, H. J. Grip strength parameters and functional activities in young adults with unilateral cerebral palsy compared with healthy subjects. Journal of Rehabilitation Medicine. 39 (8), 598-604 (2007).
- Meldrum, D., Cahalane, E., Conroy, R., Guthrie, R., Hardiman, O. Quantitative assessment of motor fatigue: normative values and comparison with prior-polio patients. Amyotrophic Lateral Sclerosis. 8 (3), 170-176 (2007).
- Schwid, S. R., et al. Quantitative assessment of motor fatigue and strength in MS. Neurology. 53, 743-743 (1999).
- Hunter, S. K., Critchlow, A., Shin, I. S., Enoka, R. M. Men are more fatigable than strength-matched women when performing intermittent submaximal contractions. Journal of Applied Physiology. 96 (6), 2125-2132 (2004).
- Karatrantou, K. Dynamic Handgrip Strength Endurance: A Reliable Measurement in Older Women. Journal of Geriatric Physical Therapy. 42 (3), 51-56 (2019).
- The National Isometric Muscle Strength Database. Muscular weakness assessment: Use of normal isometric strength data. Archives of Physical Medicine and Rehabilitation. 77 (12), 1251-1255 (1996).
- Desrosiers, J., Bravo, G., Hébert, R. Isometric grip endurance of healthy elderly men and women. Archives of Gerontology and Geriatrics. 24 (1), 75-85 (1997).
- White, C., Dixon, K., Samuel, D., Stokes, M. Handgrip and quadriceps muscle endurance testing in young adults. SpringerPlus. 2 (1), 451 (2013).
- Trajano, G., Pinho, C., Costa, P., Oliveira, C. Static stretching increases muscle fatigue during submaximal sustained isometric contractions. Journal of Sports Medicine and Physical Fitness. 55 (1-2), 43-50 (2015).
- Liu, J. Z., et al. Human Brain Activation During Sustained and Intermittent Submaximal Fatigue Muscle Contractions: An fMRI Study. Journal of Neurophysiology. 90 (1), 300-312 (2003).
- Demura, S., Yamaji, S. Influence of grip types and intensities on force-decreasing curves and physiological responses during sustained muscle contractions. Sport Sciences for Health. 3 (1), 33-40 (2008).
- Matuszczak, Y., et al. Effects of N-acetylcysteine on glutathione oxidation and fatigue during handgrip exercise. Muscle & Nerve. 32 (5), 633-638 (2005).
- Medved, I., et al. N-acetylcysteine infusion alters blood redox status but not time to fatigue during intense exercise in humans. Journal of Applied Physiology. 94 (4), 1572-1582 (2003).
- Löscher, W. N., Cresswell, A. G., Thorstensson, A. Excitatory drive to the alpha-motoneuron pool during a fatiguing submaximal contraction in man. The Journal of Physiology. 491 (1), 271-280 (1996).
- Taylor, J. L., Allen, G. M., Butler, J. E., Gandevia, S. C. Supraspinal fatigue during intermittent maximal voluntary contractions of the human elbow flexors. Journal of Applied Physiology. 89 (1), 305-313 (2000).
- Fulco, C. S., et al. Slower fatigue and faster recovery of the adductor pollicis muscle in women matched for strength with men. Acta Physiologica Scandinavica. 167 (3), 233-239 (1999).
- Gonzales, J. U., Scheuermann, B. W. Absence of gender differences in the fatigability of the forearm muscles during intermittent isometric handgrip exercise. Journal of Sports Science & Medicine. 6 (1), 98-105 (2007).
- Liepert, J., Mingers, D., Heesen, C., Bäumer, T., Weiller, C. Motor cortex excitability and fatigue in multiple sclerosis: a transcranial magnetic stimulation study. Multiple Sclerosis Journal. 11 (3), 316-321 (2005).
- Kim, J., Yim, J. Effects of an Exercise Protocol for Improving Handgrip Strength and Walking Speed on Cognitive Function in Patients with Chronic Stroke. Medical science monitor : international medical journal of experimental and clinical research. 23, 5402-5409 (2017).
- Schnelle, J. F., et al. et al Evaluation of Two Fatigability Severity Measures in Elderly Adults. Journal of the American Geriatrics Society. 60 (8), 1527-1533 (2012).
- Enoka, R. M., Duchateau, J. Translating Fatigue to Human Performance. Medicine and science in sports and exercise. 48 (11), 2228-2238 (2016).
Przedruki i uprawnienia
Zapytaj o uprawnienia na użycie tekstu lub obrazów z tego artykułu JoVE
Zapytaj o uprawnieniaThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. Wszelkie prawa zastrzeżone