Method Article
A Streamlined Approach for Mass Spectrometry-Based Proteomics Using Selected Tissue Regions
In This Article
Summary
Here, we establish a mass spectrometry-based proteomic method using isolated regions of interest in formalin-fixed, paraffin-embedded tissue sections. This protocol is used to analyze proteome from specific tissue areas in archived formalin-fixed, paraffin-embedded tissue sections.
Abstract
Mass spectrometry (MS)-based proteomics enables comprehensive proteome analysis across a wide range of biological samples, including cells, tissues, and body fluids. Formalin-fixed, paraffin-embedded (FFPE) tissue sections, commonly used for long-term archiving, have emerged as valuable resources for proteomic studies. Beyond their storage benefits, researchers can isolate regions of interest (ROIs) from normal tissue regions through collaborative efforts with pathologists. Despite this potential, a streamlined approach for proteomic experiments encompassing ROI isolation, proteomic sample preparation, and MS analysis remains lacking. In this protocol, an integrated workflow that combines macrodissection of ROIs, suspension trapping-based sample preparation, and high-throughput MS analysis is presented. Through this approach, the ROIs of patients' FFPE tissues, consisting of benign serous cystic neoplasms (SCN) and precancerous intraductal papillary mucinous neoplasms (IPMN) diagnosed by pathologists, were macrodissected, collected, and analyzed, resulting in high proteome coverage. Furthermore, molecular differences between the two distinct pancreatic cystic neoplasms were successfully identified, thus demonstrating the applicability of this approach for advancing proteomic research with FFPE tissues.
Introduction
For decades, surgically excised human tissues have been archived as formalin-fixed, paraffin-embedded (FFPE) blocks. These tissue blocks initially preserve the three-dimensional (3D) structure embedded in paraffin. The tissues are subsequently sliced using microtomes, mounted onto slides, and stained-commonly with hematoxylin and eosin (HE) or immunohistochemistry (IHC)-to facilitate histopathological diagnosis by experienced pathologists1,2. FFPE tissues offer distinct advantages for long-term storage due to the protein crosslinking induced by formalin, which halts enzymatic and proteolytic activity3. Because they support the construction of large, well-archived sample sets, FFPE tissues have been regarded as a cornerstone for biomarker discovery across diverse fields, including genomics4,5,6,7.
However, their application in liquid chromatography-mass spectrometry (LC-MS)-based proteomics has historically posed challenges. A key limitation is the formalin-induced protein crosslinking, which interferes with tryptic digestion -- a critical step in global proteome analysis8. Furthermore, the small amount of protein retrievable from tissue slides often renders conventional sample preparation methods unsuitable. Despite these challenges, advances have demonstrated that protein crosslinks can be reversed through prolonged high-temperature treatment9,10,11. Concurrently, sample preparation methods optimized for low-amount protein samples have expanded the use of FFPE tissues in proteomics research12,13,14,15.
One significant advantage of using FFPE tissue slides in proteomics lies in their capability to enable a region-specific analysis. FFPE slides typically contain both lesions and adjacent normal tissue (ANT). Analyzing the entire tissue indiscriminately risks confounding results due to mixed molecular signatures. In contrast, isolating and analyzing regions of interest (ROIs) -- lesions versus ANT -- enables more precise characterization of molecular features specific to pathological regions. Consequently, FFPE-based approaches have become increasingly popular in proteomics studies16,17,18,19,20. Despite their growing application, a streamlined workflow that describes the whole proteomic experiment step-by-step still remains scarce. In particular, a video-based protocol has not been published.
In this study, a robust LC-MS-based proteomics workflow tailored for the accurate profiling of molecular changes within lesion-specific regions was established. Using FFPE tissues diagnosed by two pathologists, the ROIs from benign serous cystic neoplasms (SCN) and precancerous intraductal papillary mucinous neoplasms (IPMN) were macrodissected, collected, and analyzed. The protocol incorporates a macrodissection of ROIs, suspension trapping-based sample preparation optimized for minimal protein inputs, and narrow-range data-independent acquisition (DIA)-MS analysis. This method enabled the identification of over 9,000 proteins from tissue areas approximately 1 cm², deciphering distinct proteomic signatures associated with SCN and IPMN.
Protocol
This study was reviewed and approved by the Institutional Review Board of Seoul National University Hospital (IRB No. 1904-114-1028). All participants provided written informed consent to participate in the study. Detailed information on all materials used in this protocol is presented in the Table of Materials.
1. FFPE tissue antigen retrieval for proteomics sample preparation
NOTE: Ensure that the scalpels and all materials, such as the tube used, are sterile to avoid any cross-contamination. Protocols of this study can be adapted for any FFPE tissue with minor modifications based on the laboratory setup.
- Cut 10-µm FFPE tissue sections from whole-mount FFPE tissue blocks and place them onto glass slides.
NOTE: In this study, human pancreatic cyst FFPE tissue (two different types of cystic tissue-SCN and IPMN) was used for proteomics sample preparation. - Remove paraffin wax of FFPE tissue (deparaffinize) with two washes in xylene with caution: One for 5 min and another for 2 min.
- Rehydrate the tissue through a serial wash with graded ethanol mixture: 3 min each in 100%, 85%, 70%, and 50% ethanol, followed by a rinse in deionized water.
NOTE: Ensure xylene and ethanol are handled in a chemical fume hood.
2. FFPE tissue protein extraction
- Prepare an HE- or IHC-stained tissue slide on which ROI is indicated by pathologists.
- Place the unstained tissue slide and the stained tissue slide with their back sides facing each other, aligning them together. Ensure ROI is observed through the glass slide.
- Remove non-interest tissue regions using the scalpel.
NOTE: If the adjacent normal tissue should be analyzed, collect them in the other tubes. - Scrape the tissue ROIs to the center of the slide using the scalpel and transfer them into the clean 1.5-mL low protein binding tubes.
- Add 180 µL of sodium dodecyl sulfate (SDS) lysis buffer (5 % SDS, 2 mM tris (2-carboxyethyl)phosphine) [TCEP], 300 mM pH 8.5 Tris-HCl in deionized water) to each tube.
NOTE: Ensure the volume of lysis buffer is sufficient to solubilize the tissue during sonication and to suspend the extracted proteins. Insufficient buffer can lead to incomplete tissue lysis, while excessive buffer may dilute the protein lysate. - Perform probe sonication at an amplitude of 20%, applying 10 cycles of 5 s on and 5 s off.
- Incubate the samples at 100 °C for 3.5 h with 1,000 x g.
NOTE: Perform water sonication of the samples for 1 min every hour during the incubation for efficient protein extraction. - After incubation, cool the sample at room temperature (RT) for 10 min. Centrifuge the samples at 16,000 x g for 10 min at RT to separate tissue debris from the supernatant.
- Collect the supernatant into a clean, labeled tube and store it at -80 °C until further use.
3. Protein quantification of FFPE tissue lysate
NOTE: Most bicinchoninic acid assay steps for protein quantification are based on the manufacturer's instructions with minor modifications. It is recommended that reagents be prepared according to the manufacturer's guidelines.
- Prepare bovine serum albumin (BSA) standards of varying concentration by serially diluting the stock solution (2 mg/mL of BSA). If necessary, dilute samples with a dilution factor of 20 or 40.
- Prepare the deionized water as the blank and the SDS lysis buffer as sample control.
- Pipette 9 µL of standards, blank, sample control, and samples to the center of the microplate well.
- Add 4 µL of compatibility reagent solution to the sample in each well.
- Cover the plate and mix on a plate shaker at medium speed for 1 min. After that, incubate the plate at 37 °C for 15 min.
- Add 260 µL of the BCA working reagent to each well, and then incubate the plate at 37 °C for 30 min.
- Cool the plate at RT for 3 min.
- Measure the absorbance of the wells at 562 nm on a plate reader.
- Subtract the 562 nm absorbance value of the blank from that of all samples and sample control.
- Further, subtract the 562 nm absorbance value of blank-subtracted sample control from that of all samples.
- Plot and use the standard curve to determine the protein concentration of each sample.
NOTE: Ensure that the estimated 200-250 µg of proteins are extracted from three glass slides of FFPE tissues, each with an approximate area of 1 cm2.
4. Acetone precipitation of protein
NOTE: Ensure that a total of 100-300 µg of protein is used for acetone precipitation and suspension trapping-based protein digestion.
- Place the protein sample (corresponding to 100-300 µg) in an acetone-compatible tube.
- Add -20 °C acetone in a volume five times the sample volume to the tube.
- Incubate the mixture at -20 °C for 18 h.
- Centrifuge the tube at 16,000 x g for 15 min.
- Carefully dispose of the supernatant without disturbing the protein pellet.
- Add 500 µL of -20 °C acetone and repeat steps 4.4-4.5.
- Air-dry the sample.
5. Suspension trapping-based protein digestion
NOTE: The suspension trapping filter-based protein digestion procedure was adapted from the manufacturer's instructions with minor modifications.
- Prepare suspension trapping lysis buffer (5 % SDS, 5 mM TCEP, 50 mM pH 8.5 TEAB in deionized water).
- Add 40 µL of the buffer to the sample tube and vortex thoroughly to dissolve the air-dried protein pellet.
- Incubate the sample at 100 °C with shaking at 1,000 x g for 35 min.
- Add 10 µL of the alkylating reagent (100 mM chloroacetamide [CAA] in 100 mM pH 8.5 Tetraethylammonium bromide [TEAB]) to the sample.
- Incubate at RT with shaking at 300 x g for 1 h.
- Prepare an acidifier (10% trifluoroacetic acid [TFA] in deionized water) with caution. Ensure that TFA, a strong acid, is handled in a chemical fume hood.
- Add 5 µL of acidifier to the sample to achieve a final concentration of 1% TFA (final sample volume: 55 µL).
- Check the pH of the sample using pH paper. Ensure the pH is less than 1.
- Add 350 µL of binding/wash buffer-1 (0.1 M pH 8.5 TEAB in 90% MeOH) to the sample to trap proteins.
NOTE: Avoid vortexing and centrifugation during the protein trapping (step 5.9) to prevent sample loss. Sufficient amounts of protein ensure the formation of visible colloidal protein particulate with a translucent appearance. - Place the suspension trapping column into a 2-mL tube and transfer the entire sample, including any insoluble material, into the suspension trapping column.
- Centrifuge the column at 4,000 x g for 50 s to trap proteins.
NOTE: Ensure all sample passes through the column; if not, repeat centrifugation until complete filtration. - Add 400 µL of the wash buffer-2 (50 % CHCl3/50 % MeOH) to the column, centrifuge at 4000 x g for 50 s, and discard the flow-through.
- Repeat step 5.12 three times.
- Add 400 µL of the buffer-1, centrifuge at 4000 x g for 50 s, and discard the flow-through.
- Repeat step 5.14 three times.
- Centrifuge at 4000 x g for 1.25 min to ensure the entire passage of buffer-1.
- Transfer the suspension trapping column to a new sample tube for enzymatic digestion.
- Add 125 µL of digestion buffer (0.16 µg/µL of trypsin/Lys-C in 50 mM pH 8.5 TEAB) to the suspension trapping column and cap it to prevent evaporation.
NOTE: The trypsin/Lys-C ratio should be 1:10 (weight/weight). Ensure a minimum of 10 µg of trypsin/Lys-C for effective digestion. - Incubate at 37 °C for 18 h without shaking.
- Prepare the following elution buffers: buffer-1 (50 mM pH 8.5 TEAB in deionized water), buffer-2 (0.2 % FA in deionized water), and buffer-3 (50 % acetonitrile [ACN] in deionized water).
- Add 80 µL of elution buffer-1 to the suspension trapping column and centrifuge at 4000 x g for 1.25 min.
- Repeat the step 5.20 with 80 µL of elution buffer-2 and buffer-3.
- Pool eluted peptides and transfer them to a clean, new tube.
6. Peptide quantification
NOTE: Most of the steps of quantitative colorimetric peptide assay are adapted from the manufacturer's instruction with minor modifications. It is recommended that reagents be prepared according to the manufacturer's guidelines.
- Prepare a series of standards by serially diluting the stock solution of the standard (1 mg/mL). If needed, dilute samples using a dilution factor of 4 or 5.
- Prepare the deionized water as the blank and a mixture of elution buffer-1, buffer-2, and buffer-3 in equal proportions as sample control.
- Place 20 µL of standards, blank, sample control, and samples into the center of each microplate well.
- Add 180 µL of the working reagent to each well and mix the plate thoroughly on a plate shaker for 1 min.
- Cover the plate and incubate the plate at 37 °C for 20 min.
- Allow the plate to cool at RT for 3 min.
- Measure the absorbance of the wells at 480 nm using a plate reader.
- Subtract the 480 nm absorbance value of the blank from that of all samples and sample control.
- Further, subtract the 480 nm absorbance value of blank-subtracted sample control from that of all samples.
- Plot and use the standard curve to calculate the peptide concentration for each sample.
7. Liquid chromatography-mass spectrometry analysis
- After peptide quantification, lyophilize 20 µg of peptides.
- Redissolve peptides in 40 µL of aqueous buffer (3 % ACN in 0.1 % formic acid [FA] water) for 10 min in a sonication bath. The resulting peptide concentration will be 0.5 µg/ µL.
- Centrifuge the sample at 16,000 x g for 60 min and transfer the samples to vials for MS analysis.
- Inject 2 µL of each sample using the autosampler of the nano LC-MS/MS system.
- Separate peptides on a reversed-phase column (0.075 mm ID x 150 mm column, packed with 3 µm C18 material, using a 127-min gradient of 5%-35% acetonitrile at 100 nL/min. Ionize the peptides via a nanospray ion source and transfer them into an orbitrap-based mass spectrometer.
- Analyze peptides using a narrow-range data-independent acquisition method21.
- Apply the following MS parameter setting: MS m/z range: 495-745, resolution target: 15000, AGC target: 3 x 10E6, max injection time: auto, MS/MS window: User defined (Supplementary File 1), resolution target: 45000, AGC target 3 x 10E6, max injection time: auto, HCD Collision Energy: 22%, 26%, 30%.
NOTE: It is recommended to perform an initial quality check of the instrument by analyzing standard samples before analysis. Use the MS data acquisition software to configure parameters (refer to Table of Materials).
8. Data analysis for proteomics search
NOTE: For proteomic search of MS raw data, open-source tools were used to convert LC-MS data format and perform proteome search (refer to Table of Materials). The parameters used for data analysis are detailed in Supplementary File 2. For basic usage instructions for open-source tools, refer to the link included in the Table of Materials.
- Covert the raw MS/MS spectrum files (*.raw) obtained from the LC-MS to the *.mzML format using open-source software.
- Use the format-converted raw files (*.mzML) as input for an open-source search engine to perform the proteome search.
- Initiate proteomic search based on the parameters specified in Supplementary File 2.
9. Statistical analysis
NOTE: For statistical analysis to identify differentially expressed proteins, an open-source tool was used to perform univariate analysis (e.g., Student's t-test; refer to Table of Materials). It is recommended to refer to basic usage instructions for the open-source tool via the link provided in the Table of Materials.
- Import the list of identified proteins (*.txt) into the open-source tool.
- Categorize samples into groups. For this study, two groups were designated: SCN and IPMN.
- Perform Log2 transformation of the abundance values to adjust the data distribution to approximate normal distribution.
- Apply a filtering step to remove proteins with missing values. In this study, a minimum of two valid values in each group was required for inclusion.
- Impute missing values using a normal distribution-based approach to generate a comprehensive list of quantified proteins for subsequent univariate analysis.
- Perform a Student's t-test to identify differentially expressed proteins between the two groups. In this study, Benjamini-Hochberg false discovery rate (FDR) correction was applied.
- Extract the differentially expressed proteins satisfying the following criteria: Benjamini-Hochberg FDR < 0.05 and |fold- change| ≥ 2.
10. Bioinformatics analysis
NOTE: A commercial bioinformatics tool was used for over-representation analysis (e.g., Ingenuity pathway analysis, refer to Table of Materials). Before using this tool, it is recommended to refer to the manufacturer's instructions.
- Import the list of DEPs (*.txt) into the commercial tool.
- Initiate core analysis to conduct over-representation analysis.
- Export significant canonical pathways enriched from the DEPs using Fisher's exact test (adjusted p-value < 0.05). Identify pathways of interest that are relevant to the research.
Results
The established suspension trapping filter-based proteomics sample preparation, combined with label-free quantitation using single-shot data-independent acquisition, were applied to pancreatic cystic FFPE tissues (Figure 1). Precise ROI isolation during the FFPE tissue processing was achieved across different pancreatic cystic FFPE tissues (Figure 2A), resulting in the acquisition of reproducible total ion chromatograms between biological tri-replicates for each type of pancreatic cystic neoplasms (Figure 2B). LC-MS analysis identified 9,703 proteins and quantified an average of 7,886 and 8,273 proteins for SCN and IPMN, respectively (Figure 2C). In addition, a total of 80,245 precursors and 75,412 peptides were identified across all six samples, with an average 55,729/51,488 and 63,573/59,519 precursors/peptides for SCN and IPMN, respectively (Figure 2D). The abundances of all of the identified proteins spanned 6.25 orders of magnitude, demonstrating comprehensive proteome coverage (Figure 2E). Furthermore, known pancreatic cancer protein markers, including KRT19, KRAS, CEACAM5, MUC1, FUT3, SPARC, SAMD4, and GATA6, which are used in prognosis and diagnosis, were impartially quantified across the entire range of proteome abundances22,23,24,25,26,27,28,29.
Pearson's correlation coefficients among the same tissue type were higher (0.92 - 0.95) than those between the tissue types (0.82 - 0.88) (Figure 2F). Principal component analysis (PCA) of SCN and IPMN showed clear grouping according to their tissue types by the first (52.2 %) and the second (15.6 %) components (Figure 2G), which corresponds to the hierarchical clustering results shown in Figure 2F. Differentially expressed proteins (DEPs) between SCN and IPMN were investigated to clarify whether biological and molecular insights could be interpreted through system-wide analysis based on our proteome. The statistical analysis identified 933 DEPs, consisting of 457 upregulated and 476 downregulated proteins in IPMN (Figure 3A). Further bioinformatics analysis revealed that the DEPs were associated with a number of pancreatic cancer-related pathways including cellular movement/proliferation-related terms -- i.e., extracellular matrix organization, integrin cell surface interactions, degradation of the extracellular matrix, and collagen degradation, cellular death-related terms -- micro-autophagy signaling pathway and intrinsic pathway for apoptosis, cell to cell signaling-related terms -- pancreatic secretion signaling pathway and ERK/MAPK signaling, and lipid metabolism-related term -- sphingolipid metabolism (Figure 3B).
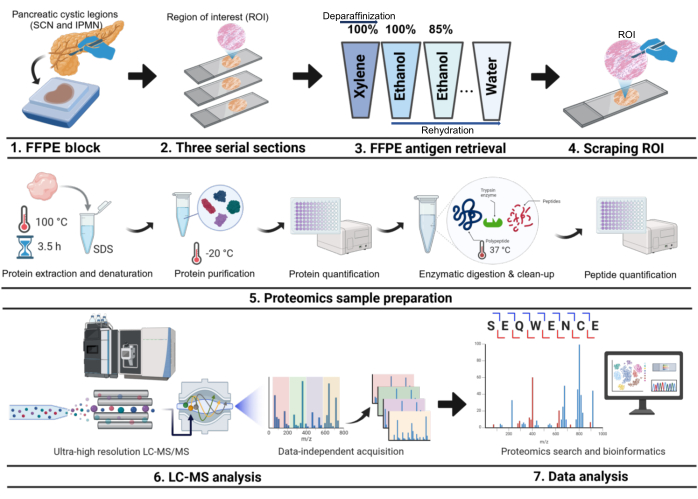
Figure 1: Experimental workflow. The workflow includes FFPE antigen retrieval, scraping ROI, protein digestion, LC-MS analysis, and data analysis. Suspension trapping filter was used in the enzymatic digestion and clean-up steps. FFPE, formalin-fixed paraffin-embedded; SCN, serous cystic neoplasms; IPMN, intraductal papillary mucinous neoplasms; LC, liquid chromatography; MS, mass spectrometry. Please click here to view a larger version of this figure.

Figure 2: Proteomic characterization of different pancreatic cystic FFPE tissues. (A) Images of FFPE tissues on the slide across samples. The region of interest (ROI) is indicated by a black line. (B) Total ion chromatogram of the samples generated by LC-MS analysis. (C) Number of identified proteins across the samples. (D) Number of identified precursors and peptides across the samples. (E) Dynamic range of the identified proteins. Known pancreatic cancer proteins are marked. (F) Pearson's correlation coefficients across the samples. Hierarchical clustering is highlighted as blue for SCN and red for IPMN, respectively. (G) Principal component analysis of different pancreatic cystic FFPE tissues based on their proteomic profiles. SCN, serous cystic neoplasms; IPMN, intraductal papillary mucinous neoplasms; BR, biological replicate. Please click here to view a larger version of this figure.
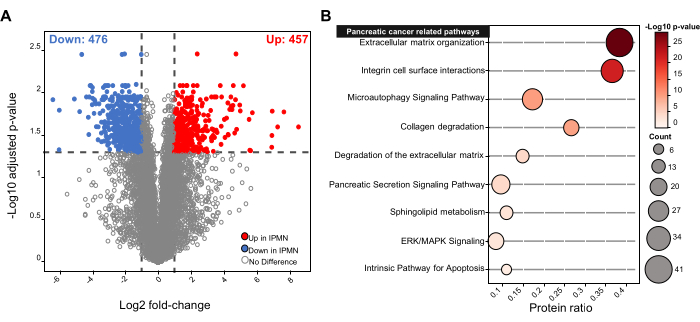
Figure 3: Proteomic alteration in different pancreatic cystic FFPE tissues. (A) Volcano plot showing differentially expressed proteins (DEPs) between IPMN and SCN (Benjamini-Hochberg FDR < 0.05 and |fold- change| ≥ 2). (B) Canonical pathways associated with pancreatic cancer were enriched from the DEPs. Protein ratio is defined as the number of proteins in a pathway that satisfies p-value < 0.05, divided by the total number of proteins in that pathway. IPMN, intraductal papillary mucinous neoplasms; FDR, false discovery rate. Please click here to view a larger version of this figure.
Discussion
This protocol outlines a rapid and efficient proteomics method that utilizes ROIs isolated from FFPE tissue sections mounted on glass slides for pathological diagnosis. When surgical intervention is advantageous, solid neoplasms such as cancers and cysts are surgically resected and preserved for pathological evaluation. For long-term storage, tissues are fixed in formalin and embedded in paraffin (FFPE). FFPE tissue blocks are then sectioned to a thickness of 4-10 µm, mounted on glass slides, subjected to antigen retrieval, and stained with HE or IHC. Pathologists evaluate cellular morphology and the expression of molecular diagnostic markers (e.g., HER2, ER, and PR) to aid in diagnostic decision-making.
Modern proteomics enables comprehensive qualitative and quantitative profiling of the human proteome in various tissues and further system-wide molecular analysis. Advances in ultra-high-resolution, ultra-fast mass spectrometers, and highly reproducible liquid chromatography have played a crucial role in these developments. Additionally, this protocol integrates sample preparation based on suspension trapping setup, which significantly reduces experimental time and label-free data-independent acquisition-mass spectrometry (DIA-MS) strategy, offering rapid and highly comprehensive proteomic profiling of ROIs of FFPE tissue section. Unlike label-based methods, label-free proteomics does not require reporter-labeling reagents, enabling better flexibility for scaling sample sets. On the other hand, protein/peptide labeling methods such as tandem mass tags (TMT) offer better protein quantification than label-free methods because they allow multiplexing of up to 35 samples, minimizing variability caused by repetitive MS analyses. However, this approach results in inefficiencies when the number of samples is fewer than or exceeds the available labeling channels. Label-free methods have been limited by unstable analytical reproducibility, low sensitivity, and reduced quantitative accuracy. Nonetheless, label-free DIA-MS has recently gained attention for its high sensitivity and deep quantitative analysis in bulk or sub-microscale samples, thereby mitigating the limitations21,30,31,32. Therefore, the label-free DIA-MS approach is particularly advantageous for in-depth quantitative proteomics analysis of large sample sets, such as archived FFPE tissues stored in biobanks or pathology departments for up to 10 years.
This method was applied to analyze proteomic differences in two distinct pancreatic cystic neoplasms, IPMN and SCN, with a focus on ROIs identified by the Department of Pathology. The LC-DIA-MS analysis identified over 9,000 proteins, more than 900 DEPs, and the pathways associated with pancreatic cancer (Figure 3). Notably, the extracellular matrix (ECM) organization and integrin cell surface interaction pathways, which have been reported to be implicated in pancreatic cancer, were predicted to be differentially activated between the two neoplasms33,34,35,36. These findings highlight molecular distinctions between precancerous IPMN and benign SCN, suggesting that this proteomics method can discover biologically relevant differences.
This protocol utilizes macrodissection for isolating ROIs, which is suitable for analyzing tumors or neoplasms with well-defined boundaries between lesion and ANT. For the cases requiring precise isolation of ROI, laser capture microdissection (LCM) is recommended, as previously reported13. However, in research settings where LCM is not accessible, this protocol provides a viable alternative for proteomic analysis when ROIs are easily distinguishable.
Disclosures
The authors have no conflict of interest to declare
Acknowledgements
All figures in this article were created with BioRender (http://www.biorender.com). This work was supported by a National Research Foundation of Korea (NRF) grants (Grant No. RS-2023-00253403 and RS-2024-00454407).
Materials
| Name | Company | Catalog Number | Comments |
| 0.1% FA in ACN (LC-MS grade) | Fisher Chemical | LS120-212 | |
| 0.1% FA in Water (LC-MS grade) | Fisher Chemical | LS118-4 | |
| 0.5M TCEP | Thermo Scientific | 77720 | |
| 10% SDS | Invitrogen | 2679093 | |
| 1M TEAB (pH 8.5) | Sigma-Aldrich | 102545001 | |
| 1M Tris-cl (pH 8.5) | BIOSOLUTION | BTO21 | |
| A-14C centrifuge | Satorious | 167709 | |
| Acetone (HPLC grade) | Fisher Scientific | A949-4 | |
| ACN (HPLC grade) | J.T.Baker | 9017-88 | |
| CHCl3 (HPLC grade) | Thermo Scientific | 022920.k2 | |
| CR paper | ADVANTEC | 70406001 | |
| DIA-NN ver 1.9 | Open source | https://github.com/vdemichev/DiaNN | Proteomics Search Engine |
| EPOCH2 microplate reader | Agilent | 2106208 | |
| Ethanol | MERCK | K50505283 836 | |
| FA (LC-MS grade) | Fisher Chemical | A117-50 | |
| Ingenuity Pathway Analysis (IPA) | QIAGEN | 830018 | Bioinformatics tool |
| Lyophilizer (SRF110R+vaper trap) | Thermo Scientific | SRF-110-115 | |
| MeOH (HPLC grade) | MERCK | UN1230 | |
| Microplate BCA protein Assay kit-Reducing Agent Compatible | Thermo Scientific | 23252 | |
| MSConvert | Open source | http://proteowizard.sourceforge.net/tools.shtml | MS data transformation software |
| Orbitrap Exploris 480 | Thermo Scientific | MA10813C | MS |
| PepMAP RSLC C18 separation column | Thermo Scientific | ES903 | |
| Perseus | Open source | https://cox-labs.github.io/coxdocs/perseus_instructions.html | Statistical tool |
| PIERCE chloroacetamide No-Weigh Format | Thermo Scientific | A39270 | |
| PIERCE Quantitative colorimetric peptide Assay | Thermo Scientific | 23275 | |
| Plate shaker | Green SSeriker | VS-202D | |
| Probe sonicator | VibraCellTM | VCX750 | |
| Protein LoBind Tube 1.5 mL | Eppendorf | 22431081 | |
| QSP 10 µL pipette Tip | Thermo Scientific | TLR102RS-Q | |
| QSP 300 µL pipette Tip | Thermo Scientific | TLR106RS-Q | |
| Scalpel | Bard-Parker | 372615 | |
| S-Trap: Rapid Universal MS sample Prep | PROTIFI | CO2-mini-40 | |
| SureSTART Vial 0.2 mL | Thermo Scientific | 6pk1655 | |
| TFA | Sigma-Aldrich | 102614284 | |
| ThermoMixer C | Eppendorf | 5382 | |
| Trypsin/Lys-C (LC-MS grade) | Promega | V5073 | |
| Vanquish NEO | Thermo Scientific | 8348249 | LC |
| Water (HPLC grade) | Honeywell | AH365-4 | |
| Xcalibur ver 4.7 | Thermo Scientific | 30966 | MS data acquisition software |
| Xylene | Sigma-Aldrich | 102033629 |
References
- Van Maldegem, F., et al. Effects of processing delay, formalin fixation, and immunohistochemistry on RNA recovery from formalin-fixed paraffin-embedded tissue sections. Diagn Mol Pathol. 17 (1), 51-58 (2008).
- Fox, C. H., Johnson, F. B., Whiting, J., Roller, P. P. Formaldehyde fixation. J Histochem Cytochem. 33 (8), 845-853 (1985).
- Fraenkel-Conrat, H., Olcott, H. S. The reaction of formaldehyde with proteins; cross-linking between amino and primary amide or guanidyl groups. J Am Chem Soc. 70 (8), 2673-2684 (1948).
- Basyuni, S., et al. Large-scale analysis of whole genome sequencing data from formalin-fixed paraffin-embedded cancer specimens demonstrates preservation of clinical utility. Nat Commun. 15 (1), 7731 (2024).
- Gracia Villacampa, E., et al. Genome-wide spatial expression profiling in formalin-fixed tissues. Cell Genomics. 1 (3), 100065 (2021).
- Zhang, X., et al. Characterization of the genomic landscape in large-scale Chinese patients with pancreatic cancer. EBioMedicine. 77, 103897 (2022).
- Ren, Z., Ordway, B., Lin, P. -. H. Archival FFPE blocks: the gift that keeps giving. Innov. 5 (1), 100532 (2024).
- Magdeldin, S., Yamamoto, T. Toward deciphering proteomes of formalin-fixed paraffin-embedded (FFPE) tissues. Proteomics. 12 (7), 1045-1058 (2012).
- Soni, R. K. Protocol for deep proteomic profiling of formalin-fixed paraffin-embedded specimens using a spectral library-free approach. STAR Protoc. 4 (3), 102381 (2023).
- Gustafsson, O. J. R., Arentz, G., Hoffmann, P. Proteomic developments in the analysis of formalin-fixed tissue. Biochim Biophys Acta Proteins Proteomics. 1854 (6), 559-580 (2015).
- O'Rourke, M. B., Padula, M. P. Analysis of formalin-fixed, paraffin-embedded (FFPE) tissue via proteomic techniques and misconceptions of antigen retrieval. Biotechniques. 60 (5), 229-238 (2016).
- Ye, X., et al. Integrated proteomics sample preparation and fractionation: method development and applications. TrAC Trends Anal Chem. 120, 115667 (2019).
- Nwosu, A. J., et al. In-depth mass spectrometry-based proteomics of formalin-fixed, paraffin-embedded tissues with a spatial resolution of 50-200 µm. J Proteome Res. 21 (9), 2237-2245 (2022).
- Coscia, F., et al. A streamlined mass spectrometry-based proteomics workflow for large-scale FFPE tissue analysis. J Pathol. 251 (1), 100-112 (2020).
- Makhmut, A., et al. A framework for ultra-low-input spatial tissue proteomics. Cell Syst. 14 (11), 1002-1014.e5 (2023).
- Shin, D., et al. Identification of TUBB2A by quantitative proteomic analysis as a novel biomarker for the prediction of distant metastatic breast cancer. Clin Proteomics. 17 (1), 16 (2020).
- Lee, H., et al. Dual oxidase 2 (DUOX2) as a proteomic biomarker for predicting treatment response to chemoradiation therapy for locally advanced rectal cancer: using high-throughput proteomic analysis and machine learning algorithm. Int J Mol Sci. 23 (21), 12923 (2022).
- Tüshaus, J., et al. Towards routine proteome profiling of FFPE tissue: insights from a 1,220-case pan-cancer study. EMBO J. 44 (1), 304-329 (2024).
- Schweizer, L., et al. Quantitative multiorgan proteomics of fatal COVID-19 uncovers tissue-specific effects beyond inflammation. EMBO Mol Med. 15 (9), e17459 (2023).
- Kobayashi, G., et al. Proteomic profiling of FFPE specimens: discovery of HNRNPA2/B1 and STT3B as biomarkers for determining formalin fixation durations. J Proteomics. 301, 105196 (2024).
- Kawashima, Y., et al. Single-shot 10k proteome approach: over 10,000 protein identifications by data-independent acquisition-based single-shot proteomics with ion mobility spectrometry. J Proteome Res. 21 (6), 1418-1427 (2022).
- Li, W., et al. Identification and prognostic analysis of biomarkers to predict the progression of pancreatic cancer patients. Mol Med. 28 (1), 43 (2022).
- Martinelli, P., et al. GATA6 regulates EMT and tumour dissemination, and is a marker of response to adjuvant chemotherapy in pancreatic cancer. Gut. 66 (9), 1665-1676 (2017).
- Racu, M. -. L., et al. Smad4 positive pancreatic ductal adenocarcinomas are associated with better outcomes in patients receiving FOLFIRINOX-based neoadjuvant therapy. Cancers. 15 (15), 3765 (2023).
- Roy, L. D., et al. MUC1 enhances invasiveness of pancreatic cancer cells by inducing epithelial to mesenchymal transition. Oncogene. 30 (12), 1449-1459 (2011).
- Shi, H., Tsang, Y., Yang, Y. Identification of CEACAM5 as a stemness-related inhibitory immune checkpoint in pancreatic cancer. BMC Cancer. 22 (1), 1291 (2022).
- Waters, A. M., Der, C. J. KRAS: the critical driver and therapeutic target for pancreatic cancer. Cold Spring Harb Perspect Med. 8 (9), a031435 (2018).
- Yao, H., et al. Glypican-3 and KRT19 are markers associating with metastasis and poor prognosis of pancreatic ductal adenocarcinoma. Cancer Biomark. 17 (4), 397-404 (2016).
- Zhan, L., Chen, L., Chen, Z. Knockdown of FUT3 disrupts the proliferation, migration, tumorigenesis and TGFβ induced EMT in pancreatic cancer cells. Oncol Lett. 16 (1), 924-930 (2018).
- Frohlich, K., Furrer, R., Schori, C., Handschin, C., Schmidt, A. Robust, precise, and deep proteome profiling using a small mass range and narrow window data-independent-acquisition scheme. J Proteome Res. 23 (3), 1028-1038 (2024).
- Li, K. W., Gonzalez-Lozano, M. A., Koopmans, F., Smit, A. B. Recent developments in data independent acquisition (DIA) mass spectrometry: application of quantitative analysis of the brain proteome. Front Mol Neurosci. 13, 564446 (2020).
- Searle, B. C., et al. Chromatogram libraries improve peptide detection and quantification by data independent acquisition mass spectrometry. Nat Commun. 9 (1), 5128 (2018).
- Prakash, J., Shaked, Y. The interplay between extracellular matrix remodeling and cancer therapeutics. Cancer Discov. 14 (8), 1375-1388 (2024).
- Perez, V. M., Kearney, J. F., Yeh, J. J. The PDAC extracellular matrix: a review of the ECM protein composition, tumor cell interaction, and therapeutic strategies. Front Oncol. 11, 751311 (2021).
- Ferrara, B., et al. The extracellular matrix in pancreatic cancer: description of a complex network and promising therapeutic options. Cancers. 13 (17), 4442 (2021).
- Weniger, M., Honselmann, K. C., Liss, A. S. The extracellular matrix and pancreatic cancer: a complex relationship. Cancers. 10 (9), 316 (2018).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved