Method Article
Quantification of Arbuscular Mycorrhizal Fungi Colonization Rate in the Study of Invasive Alien Plants
In This Article
Summary
This protocol provides a simple and easy-to-use approach for determining the colonization rate of Arbuscular mycorrhizal fungi (AMF) in the roots of invasive plants.
Abstract
Arbuscular mycorrhizal fungi (AMF) are widely distributed soil fungi in ecosystems and can form symbiotic associations (mycorrhizae) with the roots of most terrestrial plants. Plants provide carbon sources to AMF through mycorrhizal associations, while AMF hyphae can expand the range of nutrient absorption by roots and promote plant nutrient uptake. There are many different species of AMF, and the symbiotic relationships between different species of AMF and different plants vary. Invasive plants can enrich AMF species with better symbiotic capabilities through root exudates, promoting their growth and thereby increasing their colonization in invasive plant roots. At the same time, invasive plants can also disrupt the symbiotic relationship between AMF and native plants, affecting the local plant community, which is one of the mechanisms for successful plant invasion. The colonization rate of AMF in the roots of invasive and native plants indirectly reflects the role of AMF in the process of invasive plant invasion. In this method, collected plant roots can be processed directly or saved in a fixative for later batch processing. Through decolorization, acidification, staining, and destaining treatment of roots, the hyphae, spores, and arbuscular structures of AMF in the root system can be clearly observed. This method can be completed in a basic laboratory to observe and calculate the colonization rate of AMF in the root systems of invasive plants.
Introduction
Mycorrhizal fungi are prevalent in natural ecosystems and establish symbiotic relationships with the roots of most plants, forming mycorrhizae1. These associations are mutually beneficial, as plants provide photosynthetically fixed carbon compounds such as fatty acids and sugars to support the growth of mycorrhizal fungi, while the fungi reciprocate by supplying mineral nutrients like phosphorus and nitrogen to the host plants, thereby promoting plant growth2. Based on their mycorrhizal types formed with plant roots, mycorrhizal fungi can be divided into four main types: ectomycorrhizal (ECM) fungi, ericoid mycorrhizal (ERM) fungi, orchid mycorrhizal (ORM) fungi, and arbuscular mycorrhizal (AM) fungi1. Among them, arbuscular mycorrhizal fungi (AMF) have the widest distribution and can form mycorrhizal associations with over 80% of plant species3,4.
AMF play a crucial role in enhancing soil nutrient cycling5, improving plant nutrient uptake6, and regulating plant competition and succession. They play an important role in the process of invasive plant species invasion7,8. AMF, classified under the phylum Mucoromycota9, encompasses more than 250 species10. The specific symbiotic relationships between different species of AMF and different plants may vary. Invasive plant species have the potential to alter AMF diversity and promote the enrichment of AMF species with better symbiotic capabilities that contribute to their competitive advantage during growth and colonization8,11,12,13. Understanding the dynamics of AMF and their interactions with invasive plant species is essential for comprehending the mechanisms underlying plant invasions and their ecological impacts.
Qualitative studies of AMF species typically involve two main methods. One is morphological identification, such as collecting AMF spores from soil using methods like wet sieving-sucrose centrifugation, followed by classifying and quantifying the spores based on their morphology14. The other method involves molecular techniques, amplifying conserved regions of AMF genes and sequencing them for identification15. However, these methods often require extensive morphological identification experience or higher financial resources. On the other hand, quantitative studies of AMF colonization rates, while unable to determine changes in AMF species and composition, still provide a comprehensive assessment of the symbiotic relationship between AMF and plants. Such studies are indispensable in both basic research and subsequent validation work for inoculation experiments.
The colonization of AMF plays a crucial role in determining the distribution of resources among coexisting plant species7. It reflects the establishment and strength of the symbiotic relationship between AMF and host plant roots. In the same habitat, invasive plant species often exhibit higher colonization rates compared to native plants16,17. This enhanced colonization of AMF contributes to the successful invasion of invasive species such as Ambrosia artemisiifolia18, Solidago canadensis19, Sapium sebiferum20, Ageratina adenophora21, Sphagneticola trilobata22, and Flaveria bidentis7. Understanding the colonization rate of AMF in the roots of invasive plants provides a foundation for unraveling the soil microbial mechanisms underlying the successful invasion of these species. Investigating the colonization rate of AMF in invasive plant roots sheds light on the ecological implications of plant-microbe interactions and contributes to our understanding of the mechanisms driving plant invasions.
The determination of AMF colonization rate involves a staining microscopy technique, which includes several steps: root preservation, clarification, acidification, staining, destaining, and microscopic examination (Supplementary Figure 1). Over the past decades, researchers have explored various observation methods for AMF and have developed various staining techniques. In the early stages, Trypan blue staining was widely used23,24. However, this method has limitations due to the toxicity of Trypan blue. Acid fuchsin staining, on the other hand, is a commonly used method that provides bright colors, and shows reliable, and stable staining results25. Additionally, the staining solution can be reused, making it more cost-effective. The colonization rate is determined using the gridline intersect method, which provides more objective statistical results compared to other approaches26. This method is characterized by its simplicity, low cost, and minimal equipment requirements, making it feasible to be performed in basic laboratory settings. It offers a practical and accessible approach to assess the colonization rate of AMF and contributes to our understanding of the symbiotic associations between AMF and plant roots.
Protocol
We conducted experiments using one invasive plant F. bidentis and one native plant Setaria viridis. Both plants were grown in experimental plots at the Langfang Scientific Research Pilot Base of the Chinese Academy of Agricultural Sciences (CAAS), Hebei, China. Each plant species was individually planted in separate plots, with each plot measuring 2 m x 3 m and a 1-meter gap between plots. The plants were left to grow naturally, and after approximately two months, root samples were collected.
1. Root preparation and preservation
- For each plot, randomly select three plants with similar growth conditions. Loosen the soil around the plants with a shovel, and gently pull the plants out. After shaking off the soil from the roots, cut the entire root system and bring this to the laboratory. Rince the collected root samples thoroughly under running water and mix the roots from three plants within the same plot to form one replicate.
NOTE: To facilitate the description of the operational process, this protocol provides instructions for one biological replicate. To ensure the reliability of experimental results, it is recommended to collect samples from at least three biological replicates according to the experimental design during actual operations. - Use a filter paper to remove excess moisture from the cleaned roots and let them air dry.
- Use scissors to trim damaged roots, cut intact fine roots, and preserve them for storage or proceed to the next step. If proceeding with the next step within 2 days, store them in a refrigerator at 4 °C. For preserving the root samples for later use, store them in a fixative solution, such as Formalin-Aceto-Alcohol solution (FAA, 5 mL of 38% formalin + 5 mL of acetic acid + 90 mL of 70% alcohol).
NOTE: The FAA fixative is NOT mandatory. If there are only a small number of samples and can stain and count the roots within a short period after collection, skip the FAA fixative step and proceed directly after washing off the soil from the roots. When using FAA fixative, ensure that the operation is conducted in a well-ventilated area due to the presence of formaldehyde.
2. Staining of the roots
- Remove the preserved roots from the storage solution and rinse the roots clean. Place the roots in a 100 mL beaker and add about 50 mL of 10% KOH (10 g KOH + 100 mL water) solution, making sure all roots are completely immersed in the solution. Heat the beaker at 90 °C for 30 min.
NOTE: Adjust the heating time based on the maturity of the roots. This step removes pigments from the roots, making them transparent for microscopic observation. - Pour off the KOH solution from the beaker and rinse root sample gently with tap water 3x-6x to remove any remaining KOH. Pour off the excess water.
NOTE: Ensure that the beaker and all tools used are thoroughly cleaned of KOH residue, as any remnants may result in incomplete acidification in the next step, thereby affecting the staining effectiveness of the acid fuchsin. - Add a 2% HCl solution to the beaker, ensuring that the liquid completely covers the roots. Let the roots soak in the acidic solution at room temperature for 10 min.
- Discard the HCl solution from the beaker and add about 50 mL of 0.01% acid fuchsin solution (874 mL of lactic acid, 63 mL of glycerol, 63 mL of distilled water, 0.1 g acid fuchsin). Stain the roots at 90 °C for 20-60 min. After staining, the acid fuchsin solution can be recovered and reused for future staining.
NOTE: The staining time may vary depending on the age and condition of the roots, with younger and tender roots potentially requiring overnight staining at room temperature.
3. Destaining and microscopy
- Place the stained roots in about 50 mL of lactic acid (85%) solution to remove the acid fuchsin from the root cells. The separation treatment will result in AMF tissues being stained red, while root cells remain uncolored or colored light red. The central cylinder may appear red.
- Observe the root tissues and fungal tissues under a 200x microscope to ensure clear distinguishability before stopping the destaining. When the AMF structure and root cells could be clearly distinguished, the destaining process could be completed.
NOTE: Adjust the destaining time based on root maturity. During the process, select some roots for squash preparation to observe under a microscope. - Remove the roots from the lactic acid solution and transfer them to about 50 mL of glycerol (99%) solution. The acid fuchsin in the AMF structure and root cells will no longer separate out and can be used for subsequent microscopic examination.
- Place a drop of glycerol on a glass slide. For each replicate, randomly select 30-50 roots, cut 1 cm long root segments from each root. Arrange them on glass slides with glycerol, parallel to the short side of the slide, and place them parallel to each other (Supplementary Figure 1). Place 10 root sections on each slide, resulting in 3 to 5 replicates for each replicate.
NOTE: This step describes the number of root segments used for observation, referring to the number of technical replicates for each replicate. The number of root segments observed for each sample should be reasonably arranged based on the plant species and root morphology. For fine roots, observing 30-50 root segments, each 1 cm long, per sample is sufficient. However, for bigger roots, such as those of corn, it is necessary to increase the number of observations appropriately. To ensure the reliability of the statistical results, each treatment should have at least 3 biological replicates, and each biological replicate should be operated following the aforementioned method. - Cover with a coverslip. After placing the coverslip, avoid horizontally shifting the samples to prevent any rubbing or deformation of the roots. This ensures that the normal state of hyphal morphology can be observed without interference, thus maintaining the accuracy of colonization rate calculations.
- Using a 200x microscope, employ the cross-counting method to determine the colonization rate with reference to the schematic diagram (Supplementary Figure 2).
- Install a micrometer with crosshairs in the eyepiece of the microscope. Move the field of view to one end of the root and align one of the crosshairs parallel to the root. Observe whether the other line crosses with the hyphae, arbuscules, or vesicles.
- Move the field of view along the root direction to the other end, moving the same distance each time based on the coordinates of the microscope stage. Record the intersections in each field of view using a binary approach (0 or 1). If the other crosshair intersects with AMF hyphae, arbuscules, or vesicles, it is recorded as 1 in the corresponding cell of the table. Otherwise, it is recorded as 0. If there are no intersections with any of the AMF structures, it is recorded as 1 in the negative area. Observe 10 fields of view for each root segment, for a total of 100 observations are obtained per slide, resulting in 300 to 500 observations per plant sample.
4. Colonization rate calculation
- Based on the recorded numbers of intersections with hyphae (H), arbuscules (A), and vesicles (V), and non-mycorrhizal (N) in the 10 roots of each slide (as shown in Table 1), calculate the colonization rates as follows:
Hyphal colonization rate = (AH / AT) × 100%
Arbuscular colonization rate = (AA / AT) × 100%
Vesicular colonization rate = (AV / AT) × 100%
Total colonization rate = [(AT - AN) / AT] × 100%
Where:
AH: Number of intersections with hyphae
AA: Number of intersections with arbuscules
AV: Number of intersections with vesicles
AT: Total number of intersections
AN: Number of intersections with non-mycorrhizal roots
These calculations provide the colonization rates for each structure (hyphae, arbuscules, and vesicles) and the overall colonization rate, which is the sum of the colonization rates for all structures.
| Treatment | Slide | Number of intersections | ||||
| Negative | Hyphae | Arbuscules | Vesicles | Total | ||
| Sample 1 | Slide 1 | AN1 | AH1 | AA1 | AV1 | AT1 |
| Slide 2 | AN2 | AH2 | AA2 | AV2 | AT2 | |
| Slide 3 | AN3 | AH3 | AA3 | AV3 | AT3 | |
| Total | AN | AH | AA | AV | AT | |
Table 1: Statistical table of colonization rates of arbuscular mycorrhizal fungi. Abbreviations: AH = Number of intersections with hyphae; AA = Number of intersections with arbuscules; AV = Number of intersections with vesicles; AT = Total number of intersections; AN = Number of intersections with non-mycorrhizal roots.
Results
The staining results of the invasive plant roots using this method are shown in Figure 1. The structures (hyphae, arbuscules, spores, and vesicles) of AMF are stained red, the root cortex cells are stained light red after destaining, and the central cylinder is stained red. This staining result is sufficient to distinguish the fungal structures as AMF mainly exists in the plant's cortex. From the staining result, clear structures such as arbuscules (A), intraradical hyphae (IH), extraradical hyphae (EH), spores (S), vesicles (V), and hyphopodia can be observed. Arbuscules are typically located within the cortical cells of the root, exhibiting a tree-like branching structure. They are stained with a color lighter than that of hyphae and vesicles. Under a 40x objective lens, hyphae entering the cells and forming branched arbuscular structures can be observed (Figure 1B). Internal and external hyphae are found between the cells inside the root and outside the root, respectively, appearing as red or dark red filamentous structures (Figure 1). Vesicles serve as storage structures for AMF, located near the cortex or cortical cells of the root. They come in various shapes and sizes, such as circular, elliptical, or irregular spheres (Figure 1A,C). Spores, the reproductive structures of AMF, usually form in the soil. Some species may produce spores within the roots. The size and shape of spores vary, and after staining, spores appear spherical or ellipsoidal (Figure 1D). Due to the difficulty of distinguishing between internal root spores and vesicles, both are collectively counted as vesicles during statistical analysis27. The hyphopodium is a structure formed by extraradical hyphae when they reach the surface of plant roots (Figure 1E), marking the beginning of the symbiotic process as the hyphae enter the root system28.
Dark septate endophytes (DSE) are another widely distributed group of endophytic fungi that can colonize the epidermis, cortex, and vascular bundles of plant roots, forming darkly pigmented, septate hyphae and variable-shaped microsclerotia structures29. Using this staining method, the hyphal and microsclerotia structures of DSE in the root system can be simultaneously observed (Figure 1E,F). Compared to AMF, the hyphae of DSE have a darker color, ranging from deep brown to brown black or brown gray (Figure 1E). After invading the cells, DSE hyphae form swollen cells with thickened cell walls, densely packed together to form cell masses, known as microsclerotia (Figure 1F)30. Due to their distinct structures that can be observed and differentiated under a microscope, this method allows for the effective avoidance of the influence caused by DSE structures when quantifying AMF colonization rates.
Furthermore, we have also presented the summarized results of AMF colonization rates in the root systems of the invasive plant F. bidentis and native plant S. viridis (Supplementary Table 1, Figure 2). The results indicate a significantly higher total colonization rate of AMF in the root systems of the F. bidentis (P = 0.03), with both hyphal (P = 0.04) and arbuscular (P < 0.05) structures. We obtained similar results in the earlier experiments for AMF colonization rates in other invasive plants (A. artemisiifolia and Bidens pilosa) compared to native plants (S. viridis and Eclipta prostrata)8,31. The results show higher colonization rates in the invasive plant root systems than that of the native plant root systems.
It is important to note that although some AMF species produce spores inside the roots, spores are generally spherical or ellipsoidal in shape, while vesicles have an ellipsoidal or irregular shape. Therefore, it is difficult to distinguish between spores and vesicles when observing the colonization after staining. Hence, when calculating the colonization rate, we no longer distinguish between vesicles and spores; instead, we count them together as vesicles for statistical purposes.
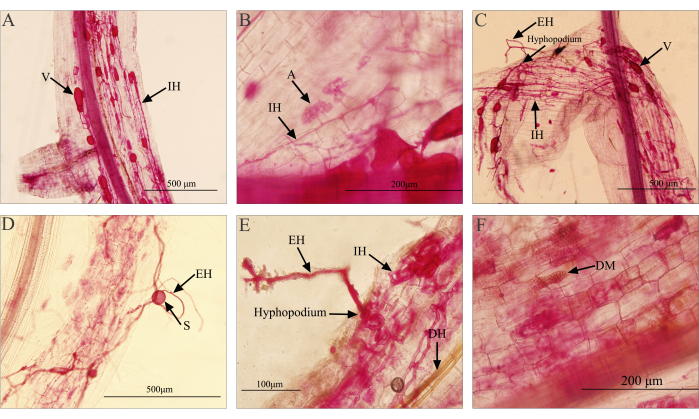
Figure 1: Microscopic examination of the mycorrhizal staining in the invasive plant Flaveria bidentis. (A, B) The images show the AMF structures observed after successful staining. (C, D) Staining results when there is improper handling, such as excessive KOH concentration during clarification or prolonged water bath time, leading to damage and peeling of the root epidermis. (E, F) Images illustrate the structures of DSE that can be observed in the root system under normal operation. Abbreviations:V = vesicles; IH = intraradical hyphae; EH = extraradical hyphae; S = spores; A = arbuscules; DH = DSE hyphae; DM= DSE microsclerotia. Please click here to view a larger version of this figure.
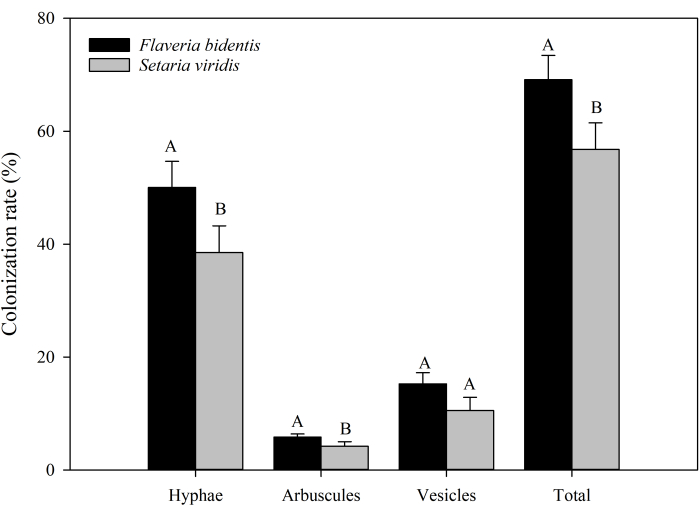
Figure 2: Colonization rates of AMF in the root systems of the invasive plant Flaveria bidentis and the native plant Setaria viridis. Different uppercase letters indicate significant differences between different plants at P < 0.05, tested by student's t test, n=3. Please click here to view a larger version of this figure.
Supplementary Figure 1: Flowchart of AMF colonization rate determination. Please click here to download this File.
Supplementary Figure 2: Schematic diagram of the method using the cross-counting method to determine the colonization rate. Revised based on the method proposed by Wang et al.32. Green rounded squares represent plant root cells, and red structures represent different structures of AMF. Circles represent the field of view of the microscope, and the crosshair within the circle indicates the crosshair line.Abbreviations:A = arbuscular, V = vesicle, and H = hypha. N = Negative. Please click here to download this File.
Supplementary Table 1: Original statistical data of AMF colonization rates in the root systems of the invasive plant Flaveria bidentis and the native plant Setaria viridis. Three replicates were assessed for each plant. Each replicate involved the preparation of three slides, with each slide counting 10 roots. In each root, 10 fields of view were observed, resulting in the examination of 300 fields of view for each replicate. Please click here to download this File.
Discussion
The interactions between invasive plants and AMF are complex and diverse. Studying these interactions is crucial for understanding the success of invasive plants and their ecological effects. They can influence the invasive ability of plants, the structure and function of soil ecosystems, and the competitiveness of native plants. The colonization rate serves as an important indicator for studying the relationship between invasive plants and AMF. It provides a quantitative measure for establishing symbiotic relationships, assessing invasive abilities, analyzing ecological effects, and studying mutual competition, thus deepening our understanding of the interactions and ecological effects between invasive plants and AMF7,8,31.
Because AMF hyphae grow inside the roots, observing their growth within the roots and quantifying the colonization rate requires root clearing and hyphal staining. Root clearing is primarily achieved using KOH, with the concentration and treatment duration adjusted as per the condition of the roots. The staining process is crucial for the successful visualization of hyphal structures. Over the past few decades, researchers have employed various staining methods, with trypan blue and acid fuchsin staining techniques being the most widely used33. In recent years, various alternative staining methods have been explored, such as methyl blue34 and ink-vinegar35. Among them, the ink-vinegar staining method is characterized by its simplicity, low toxicity, low cost, and superior staining results, leading to an increasing frequency of use. From a safety perspective, the selection of non-toxic or low-toxicity staining agents is crucial. In comparison to method using trypan blue, the acid fuchsin and ink-vinegar staining methods are considered safer and more reliable36. Considering the cost perspective, ink-vinegar is more economically feasible. However, in the case of acid fuchsin, despite its higher cost compared to ink-vinegar, its lower usage and the ability to recycle and reuse make it more suitable for laboratories engaged in long-term studies of colonization rates. Additionally, research indicates that the use of acid fuchsin staining provides better protection for the DNA integrity of AMF, facilitating subsequent identification of AMF species in root systems through molecular methods37.
There are also various methods for calculating the colonization rate, such as subjective estimation, percentage method, and weighted root segment colonization rate38,39. However, these methods are susceptible to subjective factors, resulting in variations among individuals. To address this, McGonigle et al.26 proposed the magnified intersections method, which is employed in this protocol. This method allows for more precise identification of AMF structures under high magnification microscopy and eliminates the subjective errors with estimating the degree or level of colonization. As a result, the statistical results are objective and comparable.
During these processes, there are several important considerations that need to be kept in mind, as failure to do so could lead to experiment failure:
Maintain the integrity of the root morphology
Due to the presence of AMF in the root cortex, it's important to avoid damaging the root epidermis, especially delicate fine roots. The following are key steps in this process that could potentially damage the root epidermis and result in the inability to observe infection rates (Figure 1 C and D).
Root sample collection: During field sampling, if the soil is dry and compact, it can easily damage the roots. In such cases, it's recommended to moisten the soil around the roots with water before collecting samples. If circumstances don't permit this on-site, you can excavate the roots and soil together and then moisten them in the laboratory. Peeling off the soil directly from the roots can lead to damage to the root epidermis, making it difficult to observe the infection status.
High-temperature heating: The transparent treatment and staining process are conducted at a high temperature of 90 °C. However, high temperatures can damage the structures of young and tender roots, making subsequent microscopic observation difficult. Adjust the treatment time based on the maturity of the roots, and for some cases, it may be preferable to perform the treatment at room temperature overnight.
Improper handling: Be cautious and careful throughout all procedures to avoid applying excessive force or mishandling that could damage the root surface. For instance, after root samples have undergone the clarification process and become soft, subsequent handling should avoid vigorous agitation of the samples, and the water flow during rinsing should not be too forceful. Additionally, after placing onto the slide, avoid horizontal movement of the coverslip to prevent the root system from being rubbed and deformed.
Thorough processing is essential
The following points need to be taken into consideration.
Complete the clarification treatment thoroughly: For roots with abundant pigments or older roots, if the color of most of the roots appears brown or dark yellow after the transparent treatment, it is recommended to repeat step 2.1 until the roots become colorless or semi-transparent.
Clean thoroughly: Since this method employs acid fuchsin for staining, which requires acidification of the root samples prior to staining, and given the use of a concentrated KOH solution during the earlier transparent process, it is crucial to thoroughly rinse off any residual KOH solution. Failure to do so could potentially interfere with the subsequent acidification process, resulting in incomplete acidification and, consequently, affecting the staining results. Therefore, in step 2.2, it is important to ensure a thorough cleaning of any tools that have come into contact with the KOH solution.
Determine the appropriate duration for destaining
Indeed, it is crucial to monitor the destaining process and make timely observations. In the lactic acid solution, the acid fuchsin in the root will continue to precipitate. If the destaining process takes too long, it can lead to the color being separated out from the AMF structures as well, which can affect the observation results. The destaining process should be stopped when the AMF structures can be clearly differentiated from the root structures. Due to the varying conditions of root structures among different plants, it is recommended to conduct a preliminary experiment in advance to determine the appropriate duration for the destaining process. Storing the roots in the glycerol solution can help slow down the precipitation of color in the fungal structures and preserve the staining quality for further examination.
Scientific sampling methods
To avoid obtaining data from a smaller area, appropriate sampling locations should be determined based on the size of the plot, and sampling positions should aim to cover the entire plot as much as possible. When preparing microscope slides for analysis, it is advisable to randomly select 30-50 roots and then cut 1 cm long segments from each root for individual analysis. This helps avoid cutting the same root into multiple segments for observation, as such statistical results cannot represent the entire root system.
Preliminary experiments are necessary
Although this method has a wide range of applicability, plant root conditions may vary for different plants. Therefore, it is advisable to conduct preliminary experiments before starting the actual experiment to determine the appropriate number of observations and the optimal processing time and concentration for each step.
Disclosures
Authors have nothing to disclose.
Acknowledgements
This work was funded by the National Key R&D Program of China (2021YFD1400100, 2021YFC2600400 and 2022YFC2601100), and by the National Science Foundation of China (42207162).
Materials
| Name | Company | Catalog Number | Comments |
| 70% Alcohol | Shanghai Aladdin Biochemical Technology Co., Ltd | R433197 | |
| Acetic acid solution | Shanghai Aladdin Biochemical Technology Co., Ltd | A116166 | |
| Acid fuchsin | Shanghai Aladdin Biochemical Technology Co., Ltd | A104917 | |
| Formaldehyde solution, Formalin | Shanghai Aladdin Biochemical Technology Co., Ltd | F111941 | |
| Glycerol | Shanghai Aladdin Biochemical Technology Co., Ltd | G116203 | |
| Hydrochloric acid, HCl | Shanghai Aladdin Biochemical Technology Co., Ltd | H399657 | |
| Lactic acid | Shanghai Aladdin Biochemical Technology Co., Ltd | L432769 | |
| Manual System Microscope BX43 | Olympus (China) co., Ltd | ||
| Potassium hydroxide, KOH | Shanghai Aladdin Biochemical Technology Co., Ltd | P112284 |
References
- Genre, A., Lanfranco, L., Perotto, S. Unique and common traits in mycorrhizal symbioses. Nat Rev Microbiol. 18 (11), 649-660 (2020).
- Shi, J., Wang, X., Wang, E. Mycorrhizal symbiosis in plant growth and stress adaptation: from genes to ecosystems. Annu Rev Plant Biol. 74, 569-607 (2023).
- Evelin, H., Devi, T. S., Gupta, S., Kapoor, R. Mitigation of salinity stress in plants by arbuscular mycorrhizal symbiosis: current understanding and new challenges. Front Plant Sci. 10, 470 (2019).
- Smith, S. E., Read, D. J. . Mycorrhizal symbiosis. , (2010).
- Bender, S. F., Conen, F., Heijden, M. v. d. Mycorrhizal effects on nutrient cycling, nutrient leaching and N2O production in experimental grassland. Soil Biol Biochem. 80, 283-292 (2015).
- Shen, K., et al. AM fungi alleviate phosphorus limitation and enhance nutrient competitiveness of invasive plants via mycorrhizal networks in Karst Areas. Front Ecol Evol. 8, 125 (2020).
- Zhang, F. J., et al. Arbuscular mycorrhizal fungi facilitate growth and competitive ability of an exotic species Flaveria bidentis. Soil Biol Biochem. 115, 275-284 (2017).
- Zhang, F., et al. AM fungi facilitate the competitive growth of two invasive plant species, Ambrosia artemisiifolia and Bidens pilosa. Mycorrhiza. 28 (8), 703-715 (2018).
- Spatafora, J. W., et al. A phylum-level phylogenetic classification of zygomycete fungi based on genome-scale data. Mycologia. 108 (5), 1028-1046 (2016).
- Tedersoo, L., et al. High-level classification of the fungi and a tool for evolutionary ecological analyses. Fungal Diversity. 90 (1), 135-159 (2018).
- Meinhardt, K. A., Gehring, C. A. Disrupting mycorrhizal mutualisms: a potential mechanism by which exotic tamarisk outcompetes native cottonwoods. Ecol Appl. 22 (2), 532-549 (2012).
- Dong, L. J., Ma, L. N., He, W. M. Arbuscular mycorrhizal fungi help explain invasion success of Solidago canadensis. Appl Soil Ecol. 157, 103763 (2021).
- Ramana, J. V., Tylianakis, J. M., Ridgway, H. J., Dickie, I. A. Root diameter, host specificity and arbuscular mycorrhizal fungal community composition among native and exotic plant species. New Phytol. 239 (1), 301-310 (2023).
- Mertz, S. M., Heithaus, J. J., Bush, R. L. Mass production of axenic spores of the endomycorrhizal fungus Gigaspora margarita. Trans British Mycol Soc. 72 (1), 167-169 (1979).
- Renker, C., Heinrichs, J., Kaldorf, M., Buscot, F. Combining nested PCR and restriction digest of the internal transcribed spacer region to characterize arbuscular mycorrhizal fungi on roots from the field. Mycorrhiza. 13 (4), 191-198 (2003).
- Bunn, R. A., Ramsey, P. W., Lekberg, Y. Do native and invasive plants differ in their interactions with arbuscular mycorrhizal fungi? A meta-analysis. J Ecol. 103 (6), 1547-1556 (2015).
- Majewska, M. L., Rola, K., Zubek, S. The growth and phosphorus acquisition of invasive plants Rudbeckia laciniata and Solidago gigantea are enhanced by arbuscular mycorrhizal fungi. Mycorrhiza. 27 (2), 83-94 (2017).
- Huang, D., Sang, W. G., Zhu, L., Song, Y. Y., Wang, J. P. Effects of nitrogen and carbon addition and arbuscular mycorrhiza on alien invasive plant Ambrosia artemisiifolia. Chinese J Appl Ecol. 21 (12), 3056-3062 (2010).
- Zhang, Q., et al. Positive feedback between mycorrhizal fungi and plants influences plant invasion success and resistance to invasion. PLoS ONE. 5 (8), e12380 (2010).
- Yang, Q., Li, B., Siemann, E. Positive and negative biotic interactions and invasive Triadica sebifera tolerance to salinity: a cross-continent comparative study. Oikos. 124 (2), 216-224 (2015).
- Li, L. Q., et al. Arbuscular mycorrhizal fungi enhance invasive plant, Ageratina adenophora growth and competition with native plants. Chinese J Ecol. 35 (1), 79-86 (2016).
- QI, S. S., et al. Effects of arbuscular mycorrhizal fungi on the growth and the competition of an invasive plant Wedelia trilobata. Microbiol China. 47 (11), 3801-3810 (2020).
- Phillips, J. M., Hayman, D. S. Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans British Mycol Soc. 55 (1), 158-161 (1970).
- Koske, R. E., Gemma, J. N. A modified procedure for staining roots to detect VA mycorrhizas. Mycol Res. 92 (4), 486-488 (1989).
- Kormanik, P. P., Bryan, W. C., Schultz, R. C. Procedures and equipment for staining large numbers of plant root samples for endomycorrhizal assay. Can J Microbiol. 26 (4), 536-538 (1980).
- McGonigle, T. P., Miller, M. H., Evans, D. G., Fairchild, G. L., Swan, J. A. A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol. 115 (3), 495-501 (1990).
- Souza, T. . Handbook of Arbuscular Mycorrhizal Fungi. , (2015).
- Chandwani, S., Maiti, S., Amaresan, N. . Microbial Symbionts. , (2023).
- Piercey, M. M., Graham, S. W., Currah, R. S. Patterns of genetic variation in Phialocephala fortinii across a broad latitudinal transect in Canada. Mycol Res. 108 (Pt 8), 955-964 (2004).
- Jumpponen, A. R. I., Trappe, J. M. Dark septate endophytes: a review of facultative biotrophic root-colonizing fungi. New Phytol. 140 (2), 295-310 (1998).
- Du, E., et al. Rhizoglomus intraradices and associated Brevibacterium frigoritolerans enhance the competitive growth of Flaveria bidentis. Plant Soil. 453, 281-295 (2020).
- Wang, S. Y., et al. Practical methods for arbuscular mycorrhizal fungal spore density, hyphal density and colonization rate of AMF. Bio-protocol. 101, e2104253 (2022).
- Sheng, P., Liu, R., Li, M. Methodological comparison of observation and colonization measurement of arbuscular mycorrhizal fungi. MYCOSYSTEMA. 30 (4), 519-525 (2011).
- Grace, C., Stribley, D. P. A safer procedure for routine staining of vesicular-arbuscular mycorrhizal fungi. Mycol Res. 95 (10), 1160-1162 (1991).
- Vierheilig, H., Coughlan, A. P., Wyss, U., Piche, Y. Ink and vinegar, a simple staining technique for arbuscular-mycorrhizal fungi. Appl Environ Microbiol. 64 (12), 5004-5007 (1998).
- Vierheilig, H., Schweiger, P., Brundrett, M. An overview of methods for the detection and observation of arbuscular mycorrhizal fungi in roots. Physiol Plant. 125 (4), 393-404 (2005).
- Dalpé, Y., Séguin, S. M. Microwave-assisted technology for the clearing and staining of arbuscular mycorrhizal fungi in roots. Mycorrhiza. 23, 333-340 (2013).
- Biermann, B., Linderman, R. G. Quantifying vesicular-arbuscular mycorrhizae: a proposed method towards standardization. New Phytol. 87 (1), 63-67 (1981).
- Bethlenfalvay, G. J., Pacovsky, R. S., Brown, M. S. Measurement of mycorrhizal infection in soybeans. Soil Sci Soc Am J. 45 (5), 871-875 (1981).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved